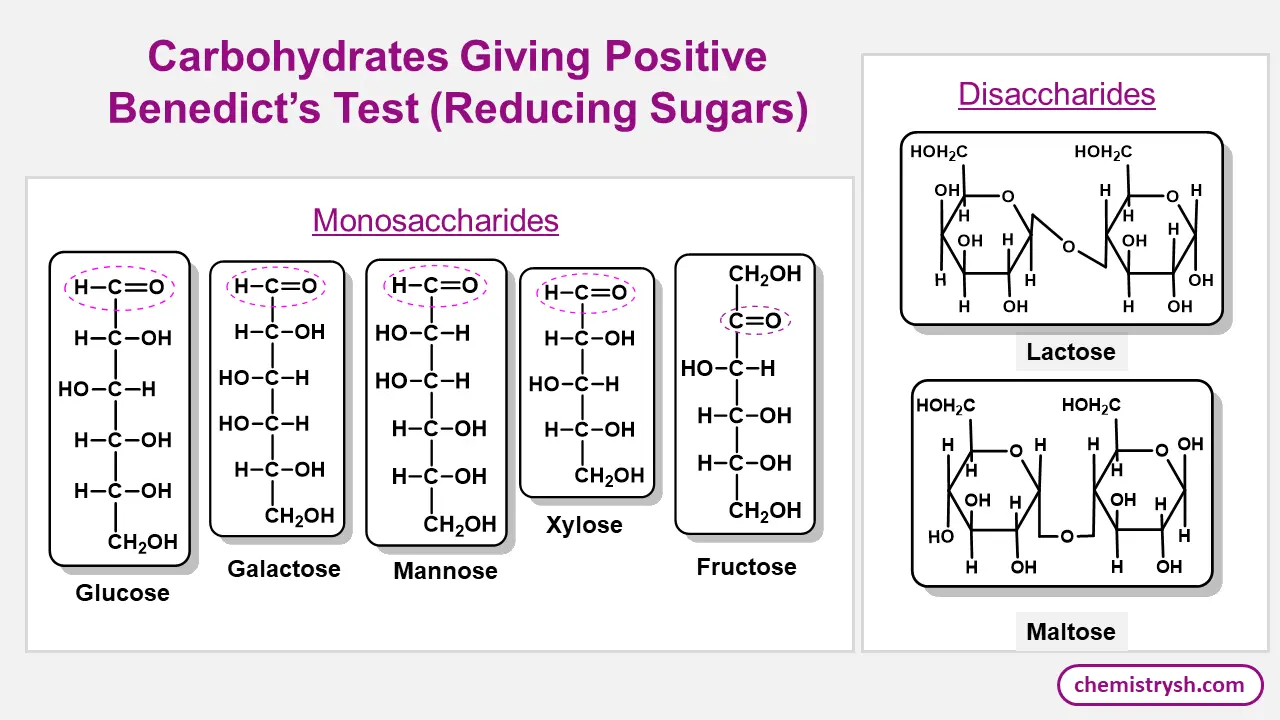
Benedict test (reducing sugar test) responds to reducing sugars like all monosaccharides (glucose, fructose, galactose, mannose), some disaccharides (maltose, lactose), certain non sugar reducing compounds (ascorbic acid (Vitamin C), uric acid, alpha hydroxy ketones) and certain drugs (e.g., salicylates, penicillin). All these compounds reduce Cu²⁺ to Cu2O in an alkaline medium used in qualitative analysis of carbohydrates in clinical and food analysis laboratories.
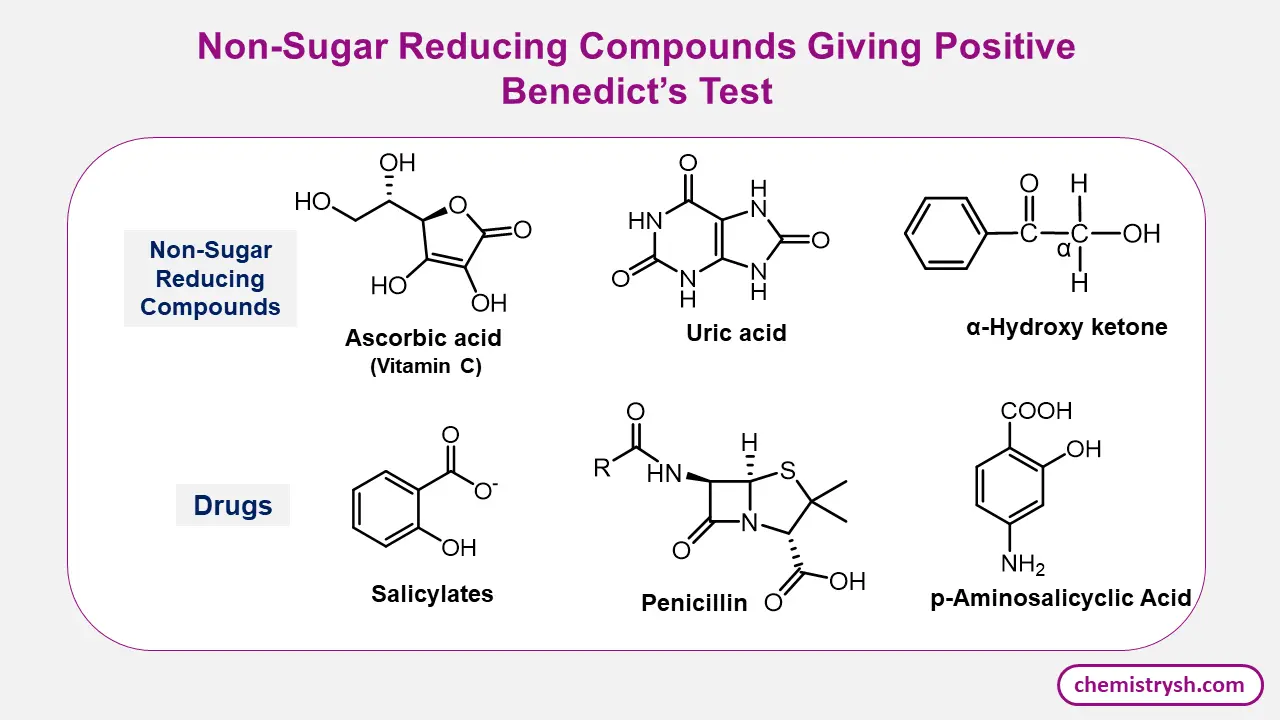
- The compounds other than sugars like non sugar reducing compounds (ascorbic acid (Vitamin C), uric acid, alpha hydroxy ketones) and certain drugs (e.g., salicylates, penicillin) can reduce the copper and give a positive result even if no sugar is present indicate the limitations of Benedict’s test.
- Benedict reagent test is also used to differentiate between aldehydes and ketones.
- Sucrose do not respond positively to Fehling solution test. After hydrolysis, such sugars may yield positive results.
What is Benedict’s Test
The Benedict’s solution test is a simple chemical and biochemical test used to find “reducing” sugars like glucose and fructose. It works through a redox reaction where the blue copper(II) ions in the solution are reduced to form a brick-red precipitate called copper(I) oxide, while the organic compound (the sugar) is oxidized.
- The Redox Process: The reaction involves the sugar donating electrons to the copper, changing the copper from a blue liquid into a solid red powder.
- Visual Color Change: You can tell how much sugar is present by the color: it starts blue (none), then turns green, yellow, orange, and finally brick-red (high sugar).
- Practical Use: This test is a staple in science labs to check for sugar in food or to help doctors screen for glucose in medical samples.
- Heat is Key: The reaction won’t happen at room temperature; the test tube must be placed in a boiling water bath for a few minutes to see the result.
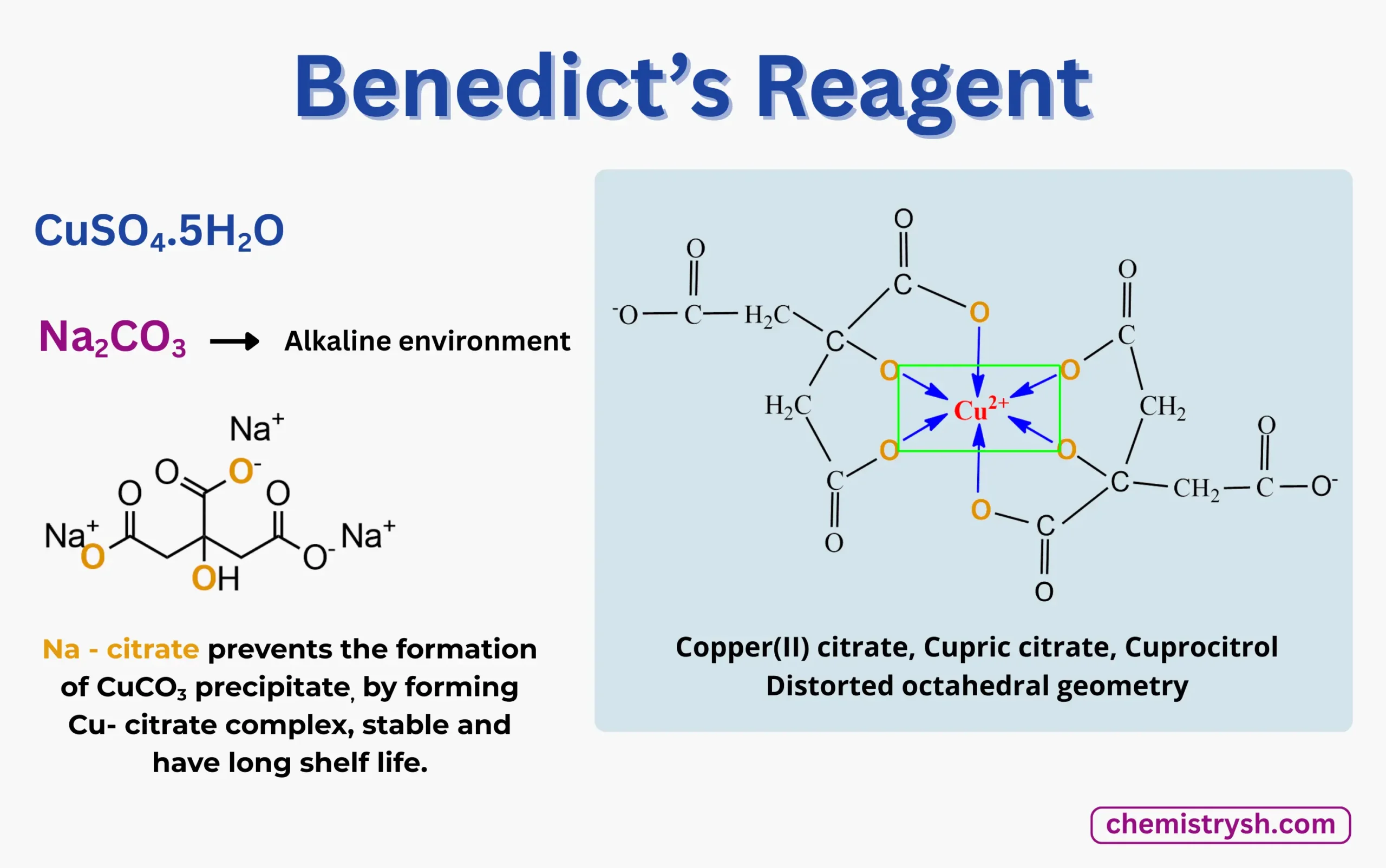
Benedict’s solution Composition / Benedict’s reagent Composition
- Copper(II) sulfate pentahydrate (CuSO₄·5H₂O) – 17.3 g
- Sodium citrate (Na₃C₆H₅O₇) – 173 g
- Sodium carbonate (Na₂CO₃) – 100 g
- Distilled water – up to 1 liter
- Beaker, stirrer, and measuring balance
Benedict’s Solution/ Reagent and Role in Reducing Sugar Test
The detection of glucose by benedict solution requires a precisely balanced reagent. Each component is essential for ensuring that the benedict’s reagent with glucose reaction produces a clear, visible color change. This composition is the standard for any food test for glucose using benedict’s solution.
|
Component |
Chemical Formula |
Amount per 1 Liter |
Function in Reducing Sugar Test |
|---|---|---|---|
|
Copper(II) sulfate pentahydrate |
CuSO₄·5H₂O |
17.3 g |
Primary Reagent: Source of Cu²⁺ ions for the redox reaction during detection of glucose. |
|
Sodium citrate |
Na₃C₆H₅O₇ |
173 g |
Stabilizer: Complexing agent that prevents copper from precipitating prematurely. |
|
Sodium carbonate |
Na₂CO₃ |
100 g |
Alkaline Medium: Provides the base needed for the detection of glucose by benedict solution. |
|
Distilled water |
H₂O |
Up to 1000 ml |
Solvent: Medium for the food test for glucose using benedict’s solution. |
Benedict’s solution Preparation / Benedict’s reagent Preparation
- Dissolve 173 g sodium citrate and 100 g sodium carbonate in 700–800 mL distilled water with continuous stirring until clear.
- Dissolve 17.3 g copper(II) sulfate pentahydrate separately in 100 mL distilled water.
- Slowly add the copper sulfate solution to the alkaline solution with constant stirring.
- Make up the final volume to 1 liter using distilled water and mix well.
- Store the prepared Benedict’s reagent in a well-sealed brown bottle at room temperature, away from direct sunlight.

Copper (II) sulphate pentahydrate/ Blue vitriol
Known for its intense blue color, blue vitriol—chemically identified as copper(II) sulfate pentahydrate—is a water-soluble crystalline compound with diverse applications. It is extensively used as a fungicide, herbicide, and algaecide, and is equally important in industrial activities such as electroplating and mining.
Notable fact
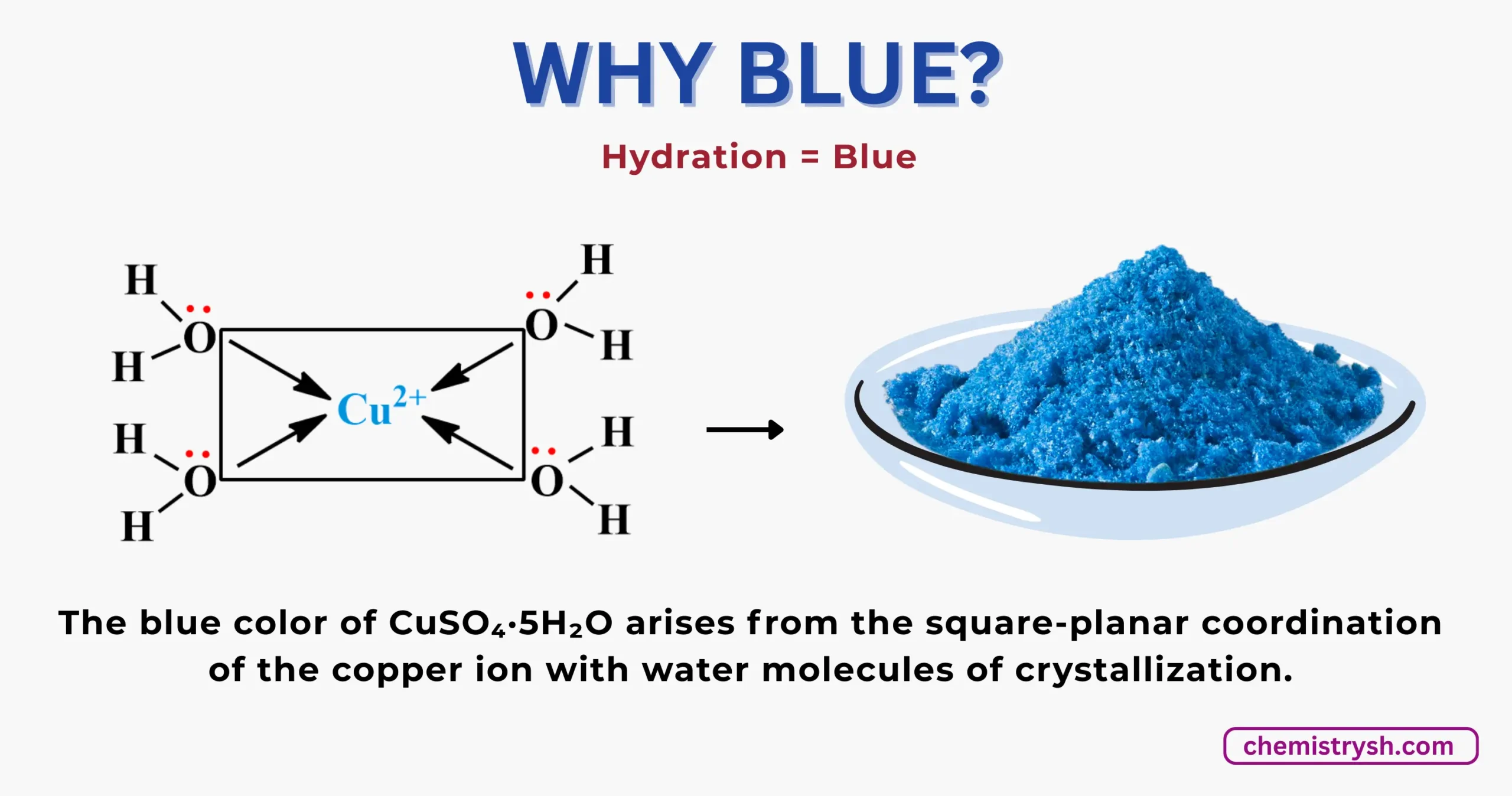
Why CuSO₄·5H₂O is blue?
Copper(II) sulfate pentahydrate, is blue because of water molecules, which make complex with the Cu2+ ions.
If we heat it, water molecules evaporate and CuSO₄ become white.
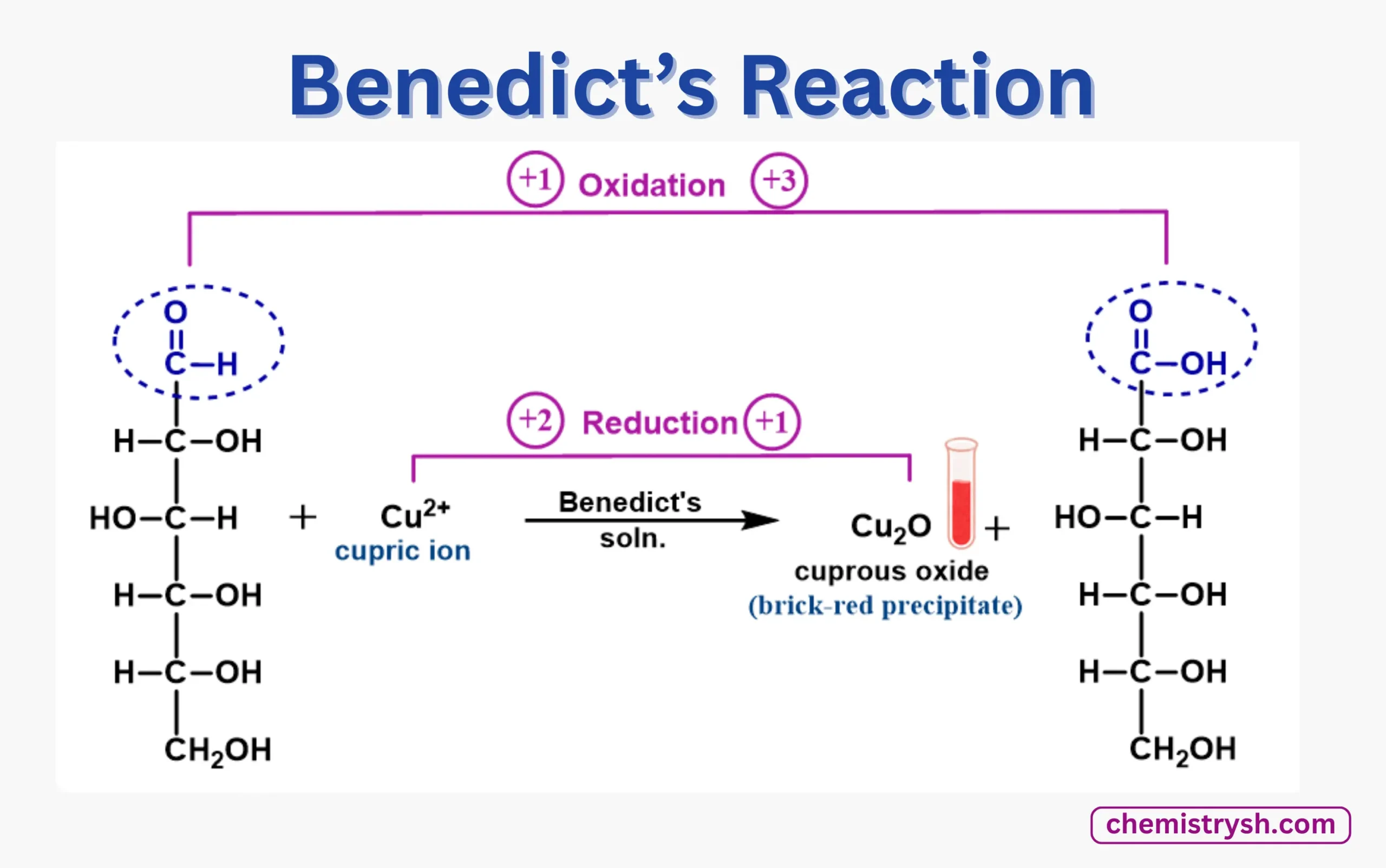
Benedict Test Principle
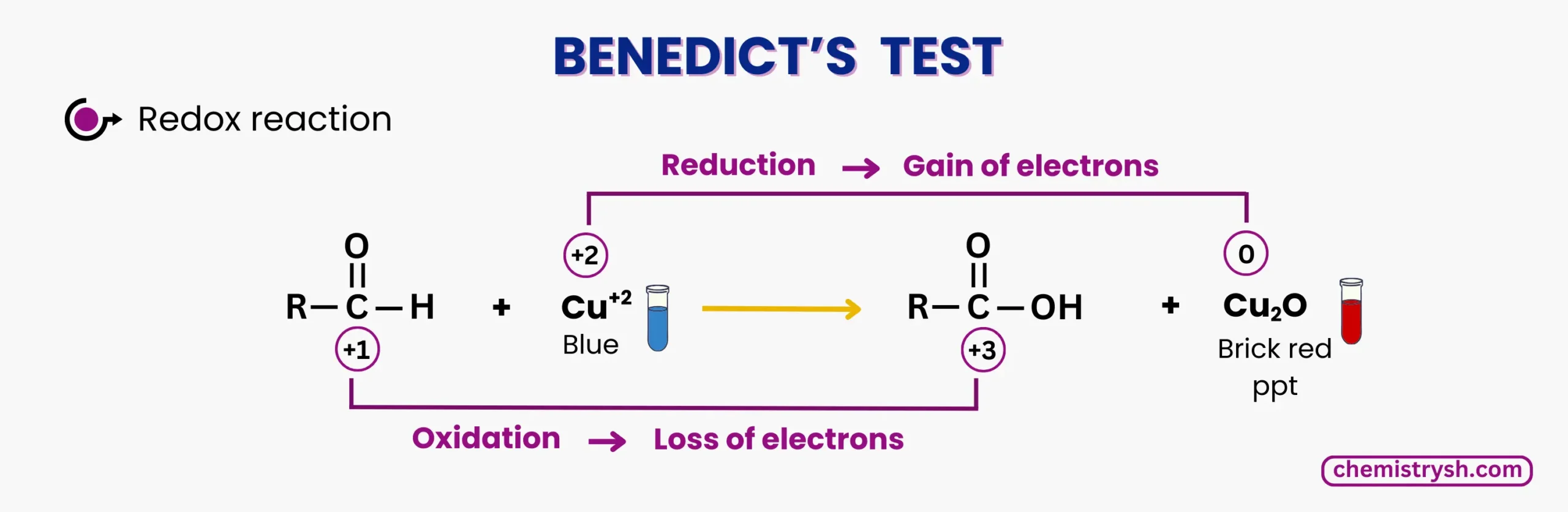
The Benedict test is based on a redox reaction between reducing sugars and copper(II) ions in an alkaline medium. Reducing sugars have a free aldehyde or ketone group that can donate electrons. During the test:
- The Cu²⁺ ions from Benedict’s reagent are reduced to Cu⁺ ions, forming copper(I) oxide (Cu₂O).
- The reducing sugar is oxidized to a carboxylic acid or carboxylate.
- The reaction produces a color change from blue (solution) to green, yellow, orange, or brick red precipitate, depending on the amount of sugar.
Benedict test Chemical Equation (Benedict glucose test):
In short: The Benedict test detects reducing sugars by oxidation of the sugar and reduction of Cu²⁺ to Cu₂O, producing a visible color change.
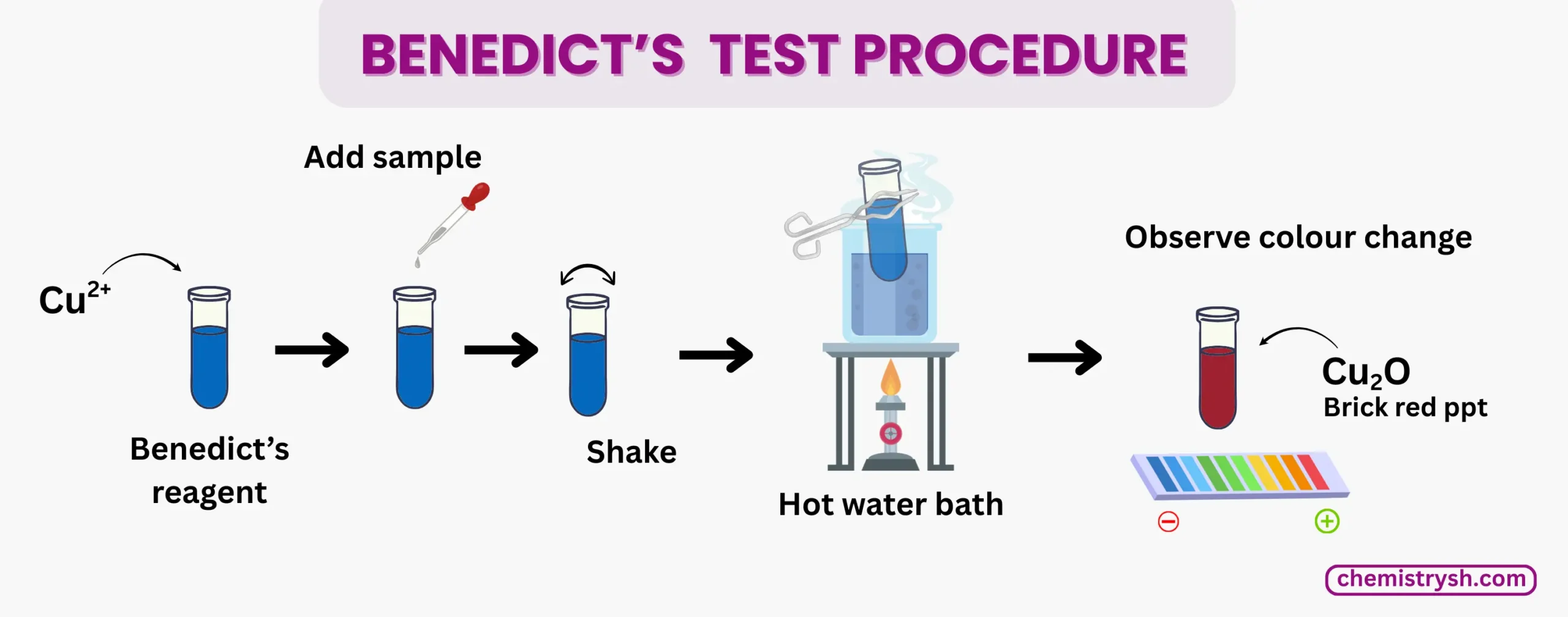
Benedict Test Procedure
- Take a clean, dry test tube, add 2-3 mL of sample solution, and dissolve solid samples in distilled water if needed
- Add 2-3 mL of Benedict’s reagent and mix gently by shaking
- Heat in boiling water bath for 3-5 minutes and observe color changes
- Remove test tube carefully and note color change and precipitate formation
Observe the Color Change in benedict test
- A blue solution indicates no reducing sugar.
- The solution may change to green, yellow, orange, or brick red precipitate, depending on the amount of reducing sugar present.
Benedict solution test Results
- No color change (remains blue): Negative, no reducing sugar.
- Green to yellow: Low concentration of reducing sugar.
- Orange to red: Moderate to high concentration of reducing sugar.

Benedict’s Test Color Observation and Interpretation Table
|
Color Observed |
Precipitate Formation |
Reducing Sugar Level |
Concentration Range |
Result Notation |
Clinical Interpretation |
|---|---|---|---|---|---|
|
Blue |
No precipitate |
Absent |
0% |
Negative (–) |
No reducing sugar detected |
|
Green |
None to slight green cloudiness |
Trace |
0.5–1.0% |
Trace (+) |
Minimal reducing sugar present |
|
Yellow |
Yellow precipitate |
Low |
1.0–1.5% |
Low (++) |
Low concentration of reducing sugar |
|
Orange |
Orange-red precipitate |
Moderate |
1.5–2.0% |
Moderate (+++) |
Moderate concentration detected |
|
Brick red |
Heavy red precipitate (Cu₂O) |
High |
>2.0% |
High (++++) |
High concentration with cuprous oxide formation |
Glucose Test
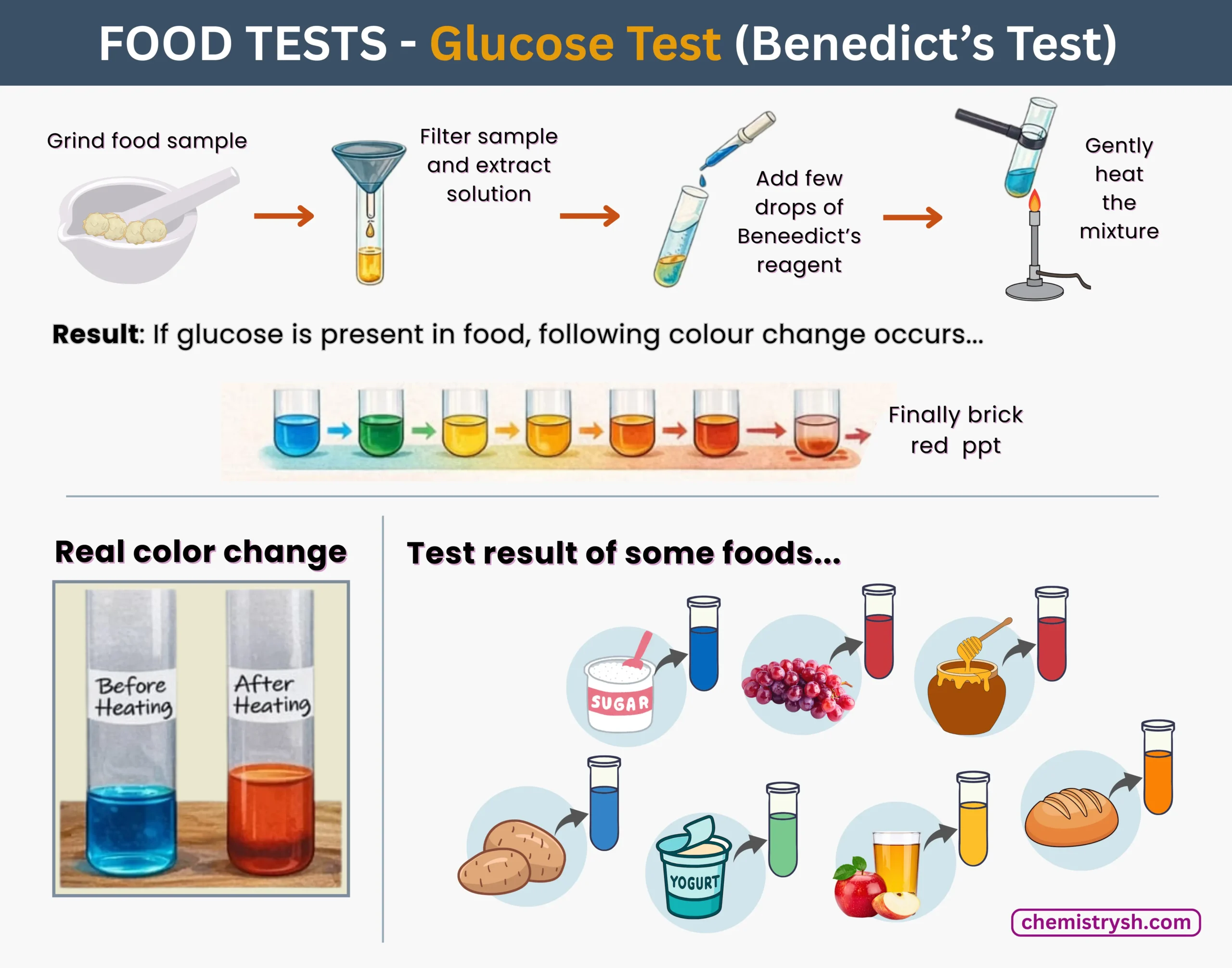
Glucose testing in foods is commonly performed to detect reducing sugars using simple laboratory methods. The presence and approximate concentration of glucose are identified by characteristic color changes after testing.
- Benedict’s test: Food samples are mixed with Benedict’s reagent and heated (~95 °C); color changes from blue to green, yellow, orange, or brick red indicate increasing glucose levels.
- Result interpretation: Blue shows no glucose, green/yellow indicates low concentration, and orange/red indicates high concentration.
- Alternative method: Glucose test strips can be used for quick testing of liquid foods such as fruit juices.
- Sample preparation: Solid or viscous foods must be crushed, diluted with water, and filtered to obtain a liquid extract for accurate results.
Benedict’s Test Chart: Results for Glucose, Sucrose, Starch, and More
Overview of Benedict’s Test Reactions This table summarizes the chemical behavior of different compound classes when subjected to benedict’s reagent with glucose and other organic molecules. It highlights the transformation of organic compounds and the visible inorganic changes used in laboratory identification. Whether you are performing a food test for glucose using benedict’s solution or the detection of glucose by benedict solution, this guide provides the expected observations.
Compounds That Respond to Benedict Test
|
Class of Compound |
Examples |
Observation with Benedict Test |
Explanation of Positive Result |
|---|---|---|---|
|
Monosaccharides |
Glucose, Fructose, Galactose, Mannose |
Brick red precipitate of cuprous oxide Cu₂O |
Free aldehyde or keto group reduces Cu²⁺ to Cu⁺ |
|
Reducing Disaccharides |
Maltose, Lactose |
Brick red precipitate of Cu₂O |
One free anomeric carbon acts as reducing end |
|
Alpha Hydroxy Ketones |
Fructose |
Brick red precipitate of Cu₂O |
Keto sugar tautomerizes to aldehyde in alkaline medium |
|
Aldehydes |
Formaldehyde, Acetaldehyde |
Brick red precipitate of Cu₂O |
Aldehyde group is easily oxidized |
|
Reducing Oligosaccharides |
Dextrins |
Brick red precipitate of Cu₂O |
Presence of free reducing ends |
|
Ascorbic Acid |
Vitamin C |
Brick red precipitate of Cu₂O |
Strong reducing agent reduces Cu²⁺ ions |
Reactivity of Ketones and α-Hydroxy Ketones in Benedict’s Test
Simple ketones generally do not give a positive Benedict’s test because they lack a free aldehyde group and are relatively resistant to oxidation under the mild alkaline conditions of the reagent.
In contrast, α-hydroxy ketones (acyloins) behave differently. In alkaline media, α-hydroxy ketones can undergo enediol formation and tautomerize into aldehyde forms. These transient aldehydes are capable of reducing cupric ions (Cu²⁺) to cuprous ions (Cu⁺), resulting in a positive Benedict’s test despite the sugar originally containing a ketone functional group.
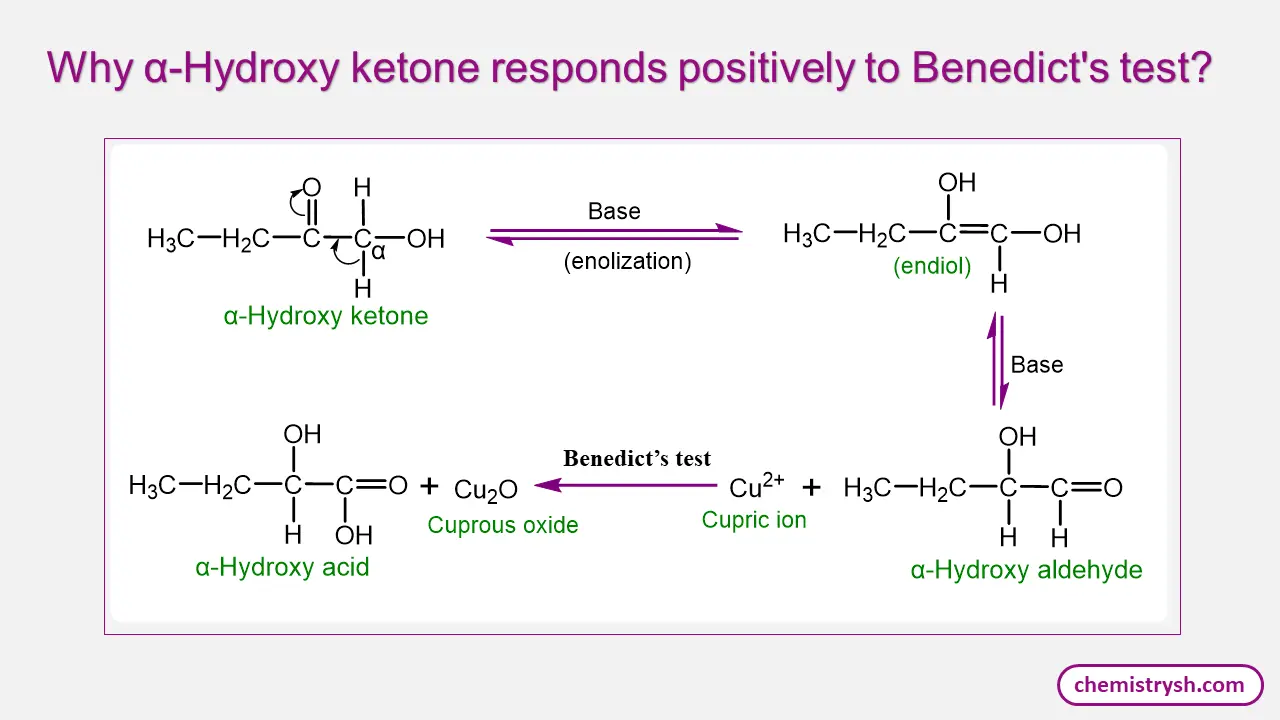
This explains why ketose sugars (e.g., fructose) still give positive results.
Key Notes for Benedict Test
The test detects reducing substances, not only sugars.
Color changes depend on concentration, from green to yellow to brick red.
Sucrose does not respond unless hydrolyzed first.
Test for non reducing sugars
Do you know?
Benedict’s test can detect all monosaccharides and some disaccharides such as lactose and maltose, but it does not react with non-reducing sugars like sucrose.
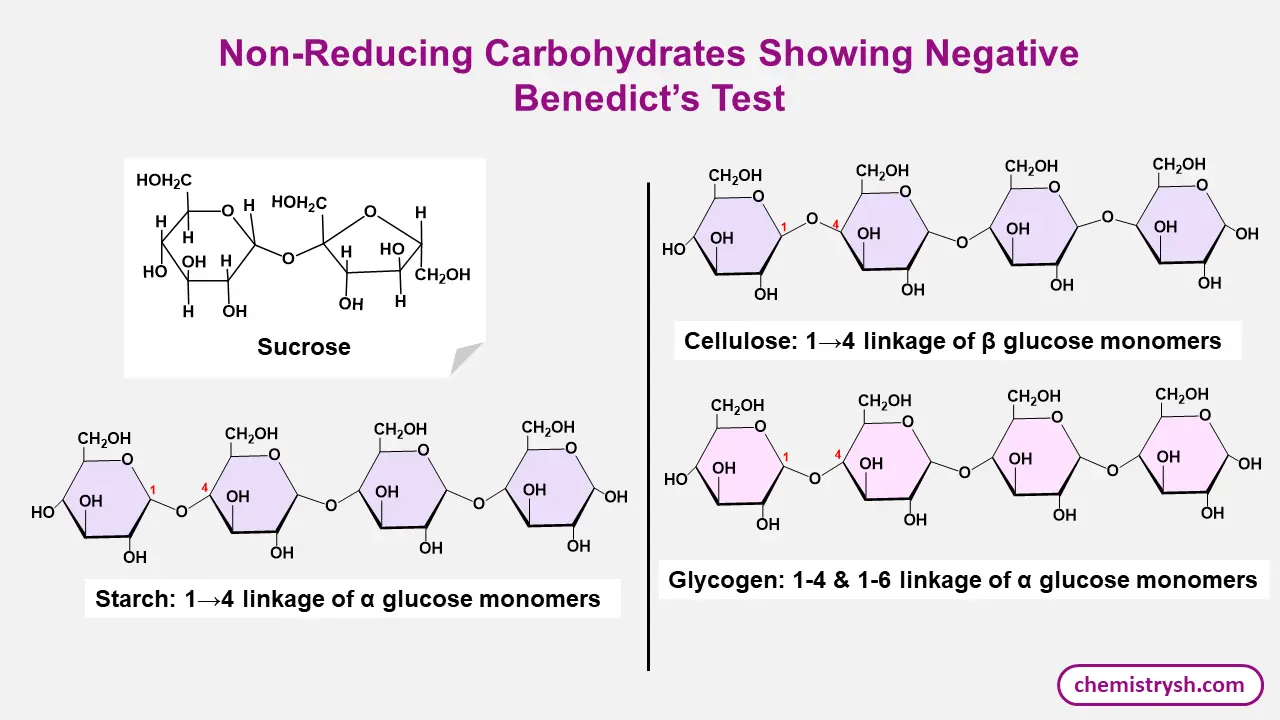
.A given sugar is confirmed as non-reducing if it gives a positive Molisch’s test, negative Benedict’s test, but becomes positive to Benedict’s test after acid hydrolysis.
- Initial Test: Perform benedict’s test on the sample. If the result is blue (negative), no reducing sugars are present.
- Hydrolysis: However, upon adding dilute hydrochloric acid to a new sample and heat in a boiling water bath for 5 minutes to hydrolyze glycosidic bonds into reducng sugars.
- Neutralization: Cool the solution and neutralize it with sodium hydrogencarbonate, Test with pH paper to ensure it is neutral or slightly alkaline, as Benedict’s reagent requires an alkaline environment.
- Re-test: Perform the Benedict’s test again by adding Benedict’s reagent and heating.
- Positive Confirmation: The shift from blue to a colored precipitate (green to brick-red) confirms the presence of non-reducing sugars.
- Common Error: Failing to properly neutralize the acid will prevent the Benedict’s reagent from working.
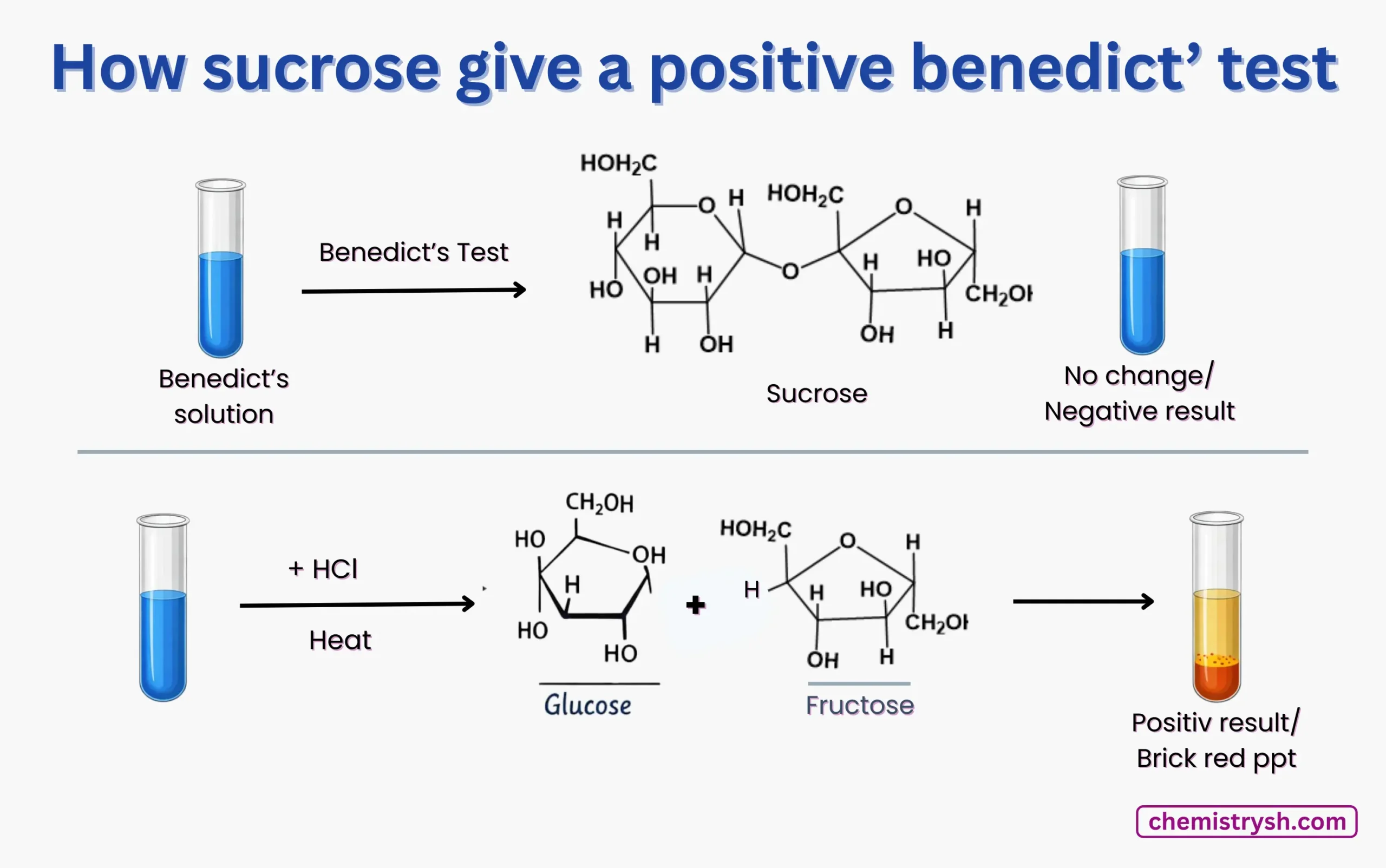
What Is Invert Sugar and Why Is It Important?
Invert sugar is produced when the non-reducing sugar sucrose undergoes hydrolysis to yield its monosaccharide components, glucose and fructose. This process, called inversion, alters the optical rotation of the sucrose to it’s component monosacchar ides and results in a mixture that is sweeter than sucrose, mainly due to the higher sweetness of fructose.
Owing to its increased sweetness, greater solubility, and reduced tendency to crystallize, this mixture is known as invert sugar or invert syrup and is widely used in food and confectionery industries.
Do you know?
Honeybees naturally produce invert sugar by using the enzyme invertase to hydrolyze nectar sucrose into glucose and fructose during honey formation. This inversion increases sweetness and prevents crystallization, giving honey its smooth texture and long shelf life.

Lactose as a Reducing Sugar in Benedict’s Test
Lactose, a reducing disaccharide, contains a free aldehyde group that can reduce metal ions. In Benedict’s test, lactose reduces the blue copper(II) ions (Cu²⁺) in Benedict’s reagent to insoluble red copper(I) oxide (Cu₂O). This red precipitate forms as a result of the oxidation of the aldehyde group in lactose, signaling a positive test.
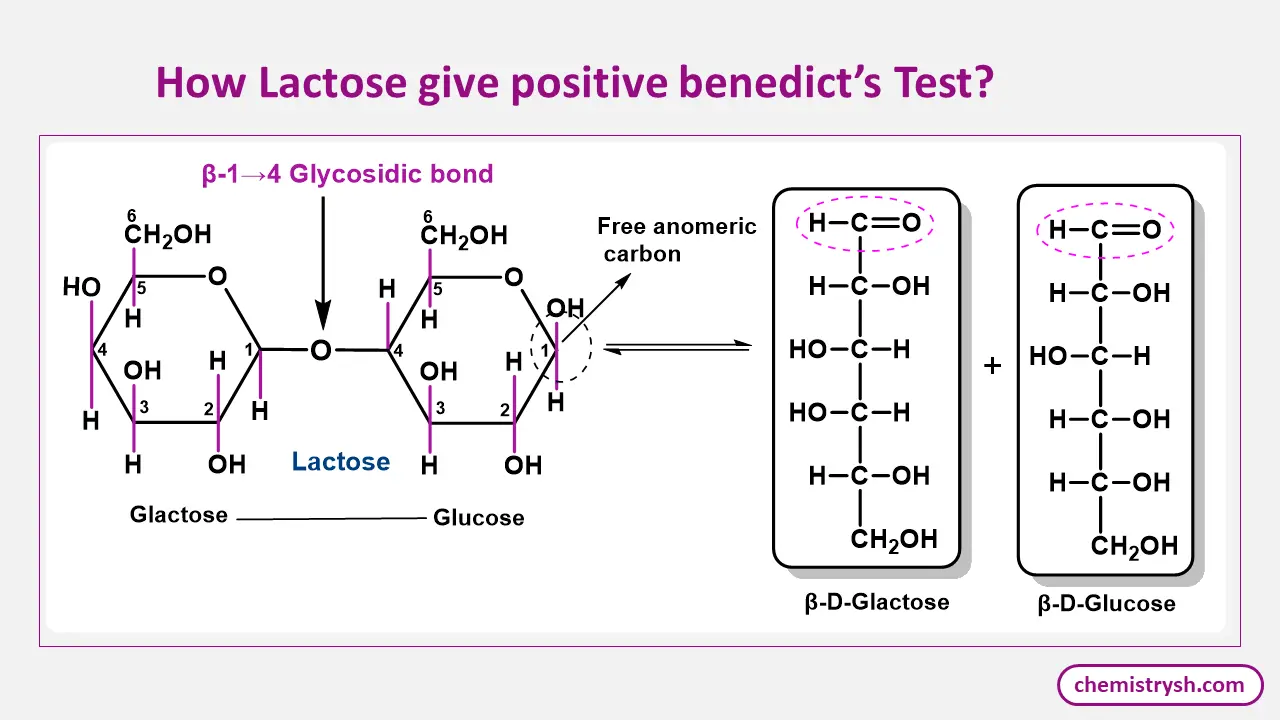
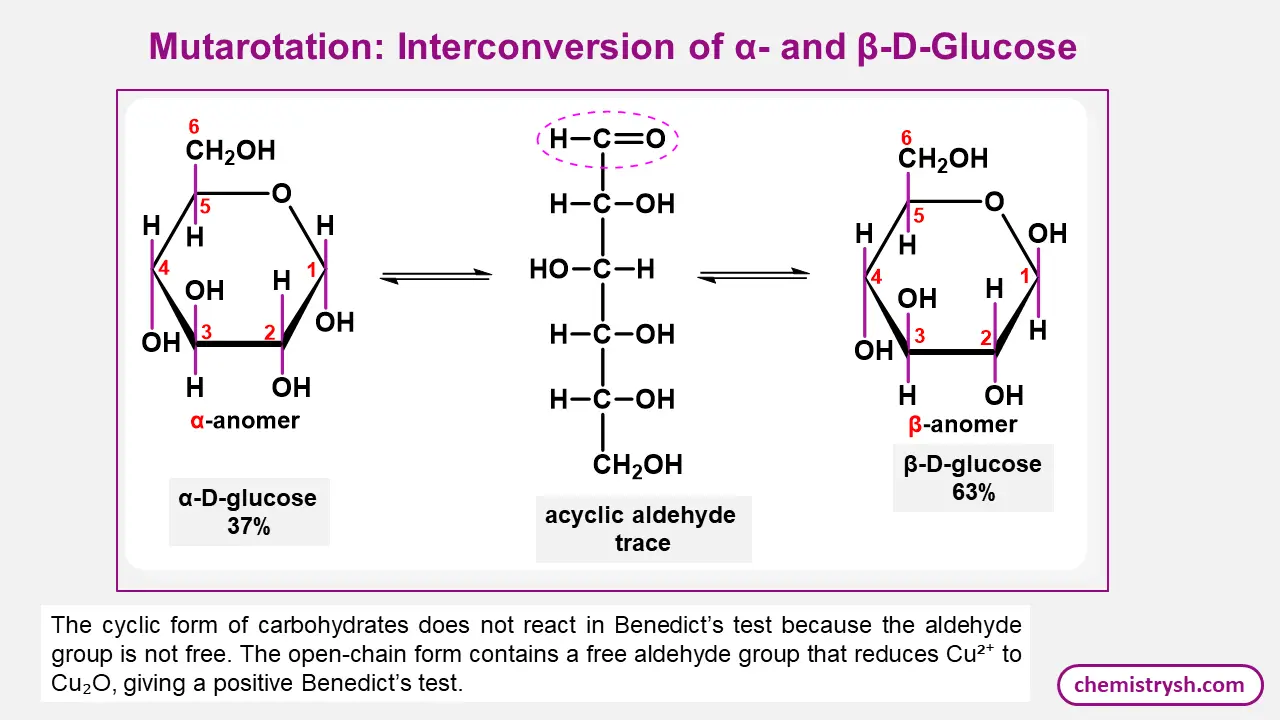
Mutarotation in Benedict’s Test
Mutarotation allows reducing sugars in aqueous solution to interconvert between their α- and β-anomeric forms, making ring opening possible. This transient formation of the open-chain aldehyde or α-hydroxy ketone structure is essential for Benedict’s test, as only these forms can reduce copper(II) ions to copper(I) ions.
The continuous establishment of equilibrium ensures the availability of the reactive species required for a positive test, even when the sugar is initially present in a single anomeric form.
- Occurs in reducing sugars due to a free anomeric carbon.
- Enables ring opening of cyclic hemiacetals.
- Generates aldehyde or α-hydroxy ketone groups.
- Provides the functional groups needed to reduce Cu²⁺ to Cu⁺.
- Maintains equilibrium in aqueous test conditions.
- Supports positive Benedict’s test results for sugars like glucose, fructose and lactose.
Objectives of the Benedict Test
- To confirm the presence of reducing sugars in an unknown sample.
- To distinguish between reducing sugars (glucose, lactose) and non-reducing sugars (sucrose).
- To estimate the concentration of sugar in a solution.
Benedict Test: Advantages and Limitations
1. Advantages of Benedict test
- Simple, cost-effective, and requires minimal equipment.
- Provides a quick visual estimate of sugar levels.
2. Limitations of Benedect test
- It is not specific to one type of sugar (it detects any reducing sugar).
- Non-reducing sugars like sucrose will give a negative result unless they are first hydrolyzed with acid.
- Certain drugs (like penicillin) or high levels of creatinine in urine can cause false positives
Benedict’s Test vs Fehling’s Solution Test
|
Aspect |
Benedict’s Test |
Fehling’s Solution Test |
|---|---|---|
|
Reagent Composition |
Single solution: copper sulfate, sodium citrate, sodium carbonate |
Two solutions mixed before use: Solution A (copper sulfate) + Solution B (Rochelle salt, sodium hydroxide) |
|
Stability |
Highly stable, can be stored for long periods |
Less stable, must be prepared fresh before use |
|
pH Condition |
Alkaline (moderately) |
Strongly alkaline |
|
Sensitivity |
More sensitive, detects lower concentrations |
Less sensitive |
|
Ease of Use |
Simple, ready-to-use single solution |
Requires mixing two solutions immediately before testing |
|
Color Change |
Blue → Green → Yellow → Orange → Brick-red precipitate |
Blue → Green → Yellow → Orange → Brick-red precipitate |
|
Principle |
Reduction of Cu²⁺ to Cu⁺ (cuprous oxide) |
Reduction of Cu²⁺ to Cu⁺ (cuprous oxide) |
|
Temperature Required |
Heating required (boiling water bath) |
Heating required (boiling) |
|
Quantitative Analysis |
Primarily qualitative |
Can be used for quantitative analysis |
|
Common Applications |
Clinical testing (urine glucose), educational labs, diabetes screening |
Historical industrial use, quantitative sugar analysis |
Key Takeaway: Both tests detect reducing sugars (glucose, fructose, maltose, lactose) but NOT non-reducing sugars (sucrose). Benedict’s test is preferred for routine use due to its stability and convenience.
Applications of Benedict’s Test
- Detection of reducing sugars
Used to identify the presence of reducing sugars such as glucose, fructose, lactose, and maltose in a sample. - Clinical diagnosis of diabetes mellitus
Helps detect glucose in urine (glycosuria), which is an important indicator of diabetes. - Semi-quantitative estimation of sugar concentration
The color change (blue → green → yellow → orange → brick-red) gives an approximate idea of the amount of reducing sugar present. - Biochemical analysis in laboratories
Commonly used in biochemistry and pathology labs for routine carbohydrate analysis. - Food and beverage testing
Applied to check the presence of reducing sugars in food products such as fruit juices, milk, and syrups. - Educational and teaching purposes
Widely used in schools and colleges to demonstrate the chemical behavior of reducing sugars. - Differentiation of carbohydrates
Helps distinguish reducing sugars from non-reducing sugars (e.g., sucrose gives a negative test unless hydrolyzed).
Multiple Choice Questions
MCQ 1
1. Which of the following disaccharides will give a NEGATIVE result with Benedict’s test?
A. 0.1% sugar concentration
B. 0.5% sugar concentration
C. 2% sugar concentration
D. 5% sugar concentration
MCQ 2
2. When testing different carbohydrates with Benedict’s reagent, which will show a positive result?
A. Starch
B. Cellulose
C. C. Glucose
D. Glycogen
MCQ 3
3. All of the following are reducing disaccharides EXCEPT:
A. Maltose
B. Sucrose
C. Lactose
D. Cellobiose
MCQ 4
4. Which of the following is NOT a limitation of Benedict’s test?
A. . It is non-specific and cannot distinguish between different reducing sugars
B. It requires heating which may affect heat-sensitive samples
C. It can detect non-reducing sugars like sucrose
D. It gives false positives with substances like vitamin C
MCQ 5
5. Benedict’s test is considered semi-quantitative because:
A. It provides exact concentration values
B. It only provides approximate concentration ranges based on color
C. It cannot detect any sugars
D. It works only in acidic conditions
MCQ 6
6. A major disadvantage of Benedict’s test compared to modern glucose detection methods is:
A. It is too expensive
B. It is too specific for glucose only
C. Color interpretation can be subjective and it takes 3-5 minutes
D. It doesn’t require any reagents
MCQ 7
7. The sensitivity of Benedict’s test for detecting reducing sugars is approximately:
A. 0.1% sugar concentration
B. 0.5% sugar concentration
C. 2% sugar concentration
D. 5% sugar concentration
MCQ 8
8. Benedict’s test has been largely replaced in clinical settings because:
A. It is more accurate than modern methods
B. Modern enzymatic glucose tests are more precise and specific
C. It is too expensive for hospitals
D. It detects only non-reducing sugars
MCQ 9
9. The accuracy of Benedict’s test is primarily limited by:
A. Its inability to detect any sugars
B. Subjective color interpretation and interference from other reducing substances
C. The requirement for special equipment
D. Its high cost
MCQ 10
10. For clinical purposes, Benedict’s test is classified as:
A. Highly quantitative and precise
B. Semi-quantitative with moderate accuracy
C. Completely inaccurate
D. Only qualitative with no concentration estimation
Viva Questions
FAQs
Conclusion of Benedict’s Test
The Benedict’s test remains a fundamental biochemical assay for detecting reducing sugars through a simple yet effective redox reaction. By reducing copper(II) ions to copper(I) oxide in an alkaline medium, this test provides a visual, semi-quantitative method for identifying monosaccharides and certain disaccharides through distinctive color changes ranging from blue to brick-red.
While it offers significant advantages including simplicity, cost-effectiveness, and minimal equipment requirements, users must be aware of its limitations, particularly its lack of specificity and susceptibility to false positives from non-sugar reducing compounds such as ascorbic acid, uric acid, and certain medications upon hydrolysis.
Complementary Tests related to Benedict’s Test
- Fehling’s Test – Confirms reducing sugars by formation of a brick-red cuprous oxide precipitate.
- Tollens’ Test – Detects aldehyde groups in reducing sugars by silver mirror formation.
- Molisch’s Test – General test to confirm the presence of carbohydrates.
- 2,4-DNPH Test – Detects carbonyl groups (aldehydes or ketones) by formation of yellow or orange
References relate to Benedict’s Test
- World Journal of Chemical Education
- Sadasivam, S., & Manickam, A. (2008). Biochemical methods (3rd ed.). New Age International.
- Nelson, D. L., & Cox, M. M. (2021). Lehninger principles of biochemistry (8th ed.). W. H. Freeman and Company.
- Murray, R. K., Bender, D. A., Botham, K. M., Kennelly, P. J., Rodwell, V. W., & Weil, P. A. (2018). Harper’s illustrated biochemistry. McGraw-Hill Education.
- Jayaraman, J. (2011). Laboratory manual in biochemistry (2nd ed.). Wiley Eastern.
- Benedict, S. R. (1909). A reagent for the detection of reducing sugars. Journal of Biological Chemistry, 5(5), 485–487.
- Benedict, S. R. (1911). The detection and estimation of reducing sugars. Journal of Biological Chemistry, 9(1), 57–59.






Thanks for finally talking about > Benedict Test for reducing sugars:
Principle, Procedure < Liked it!