The iodoform test (haloform reaction) is given by methyl ketones (CH₃CO−), acetaldehyde, ethanol, and secondary alcohols with the structure R-CHOH-CH₃. When these compounds react with iodine in the presence of a base, they produce iodoform (CHI₃), a yellow precipitate of iodoform with a characteristic antiseptic odor.

Secondary alcohols are first oxidized to methyl ketones (CH₃CO−) during the reaction, while ethanol is oxidized to acetaldehyde. Acetaldehyde (CH₃CHO) is the only aldehyde that gives a positive iodoform test.
The iodoform test originated in the 19th century and became widely adopted due to its distinctive results and simplicity. Iodoform itself has a unique penetrating antiseptic odor and was historically used as a wound disinfectant, making it easily identifiable in laboratory work.
Do you know?
During World War I, iodoform was used as an antiseptic in a paste called BIPP (Bismuth Iodoform Paraffin Paste), introduced in 1916. It was packed into infected gunshot wounds and fractures, could remain in place for days, reduced painful dressing changes, and helped prevent serious infections such as gas gangrene.
Learning Outcomes of the Iodoform Test
After studying the Iodoform Test students will be able to:
- Identify functional groups: Recognize compounds containing methyl ketones (CH₃–CO–) or secondary alcohols (CH₃–CH(OH)–) that give a positive test.
- Understand reaction mechanism: Explain the steps of the iodoform reaction, including halogenation, oxidative C–C cleavage, and carboxylate formation.
- Explain the role of OH⁻: Describe how hydroxide ions act as both a base and a nucleophile in the reaction.
- Observe characteristic changes: Identify the formation of yellow iodoform crystals (CHI₃) and understand their significance.
- Differentiate compounds: Distinguish methyl ketones, ethanol, and secondary alcohols from other carbonyl compounds and alcohols.
- Apply in qualitative analysis: Use the iodoform test to systematically detect and confirm functional groups in unknown organic compounds.
- Relate to real-life applications: Understand the historical medicinal use of iodoform and its relevance in organic chemistry education.
What is the Iodoform Test?
The iodoform test is used for the identification of methyl ketones, secondary alcohols that oxidize to methyl ketones, ethanol, and acetaldehyde. These compounds react with iodine in the presence of hydroxide ions through an oxidation and cleavage mechanism.

During the reaction, the methyl ketone is oxidized and cleaved at the carbonylcarbon and methyl carbon bond. The methyl group is converted into iodoform (CHI₃) also known as triiodomethane. The remaining carbonyl fragment forms a carboxylate ion in the basic medium.
The appearance of pale yellow iodoform crystals with a characteristic antiseptic odor serves as a positive indicator. This observation makes the iodoform test a reliable tool for identifying specific structural features in unknown organic compounds.
Why iodoform has a characteristic antiseptic odor?
Iodoform (CHI₃) has a very low odor threshold due to its heavy iodine atoms, which increase molecular volatility. Even small amounts release iodine vapors, making the smell detectable before the yellow crystals are clearly visible.
Iodoform Test Reagents
- I₂ – Iodine solution: Provides halogen for the reaction
- NaOH – Sodium hydroxide: Creates alkaline medium
- H₂O – Distilled water: Dissolves and dilutes reagents
- Gentle heat: Helps form yellow iodoform crystals
- Compound to be tested.

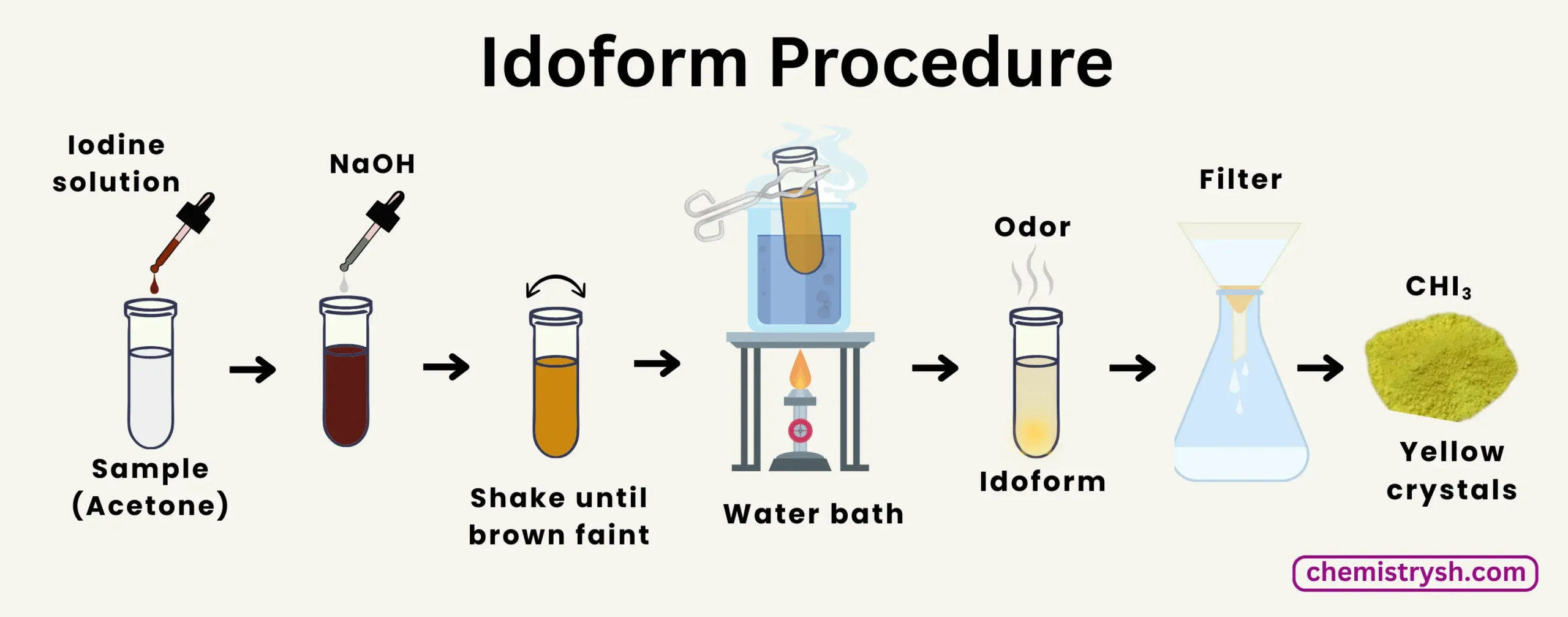
Iodoform Test Procedure
- Place 2–3 mL of the test compound in a clean test tube.
- Add ~2 mL of 10% NaOH solution.
- Add iodine dropwise with constant shaking until a faint brown color remains.
- Gently warm the test tube in a water bath (60–70°C) for a few minutes.
- Observe for yellow CHI₃ crystals and a distinct antiseptic odor, indicating a positive test.
Iodoform Test Is Given By
- Methyl ketones (CH₃CO−): Acetone (CH₃COCH₃), Acetophenone (C₆H₅COCH₃), Methyl ethyl ketone (CH₃COC₂H₅)
- Aldehyde: Acetaldehyde (CH₃CHO)
- Primary alcohol: Ethanol (CH₃CH₂OH)
- Secondary alcohols: Isopropyl alcohol (CH₃CHOHCH₃), 2-Butanol (CH₃CH(OH)CH₂CH₃), 2-Pentanol (CH₃CH(OH)CH₂CH₂CH₃)
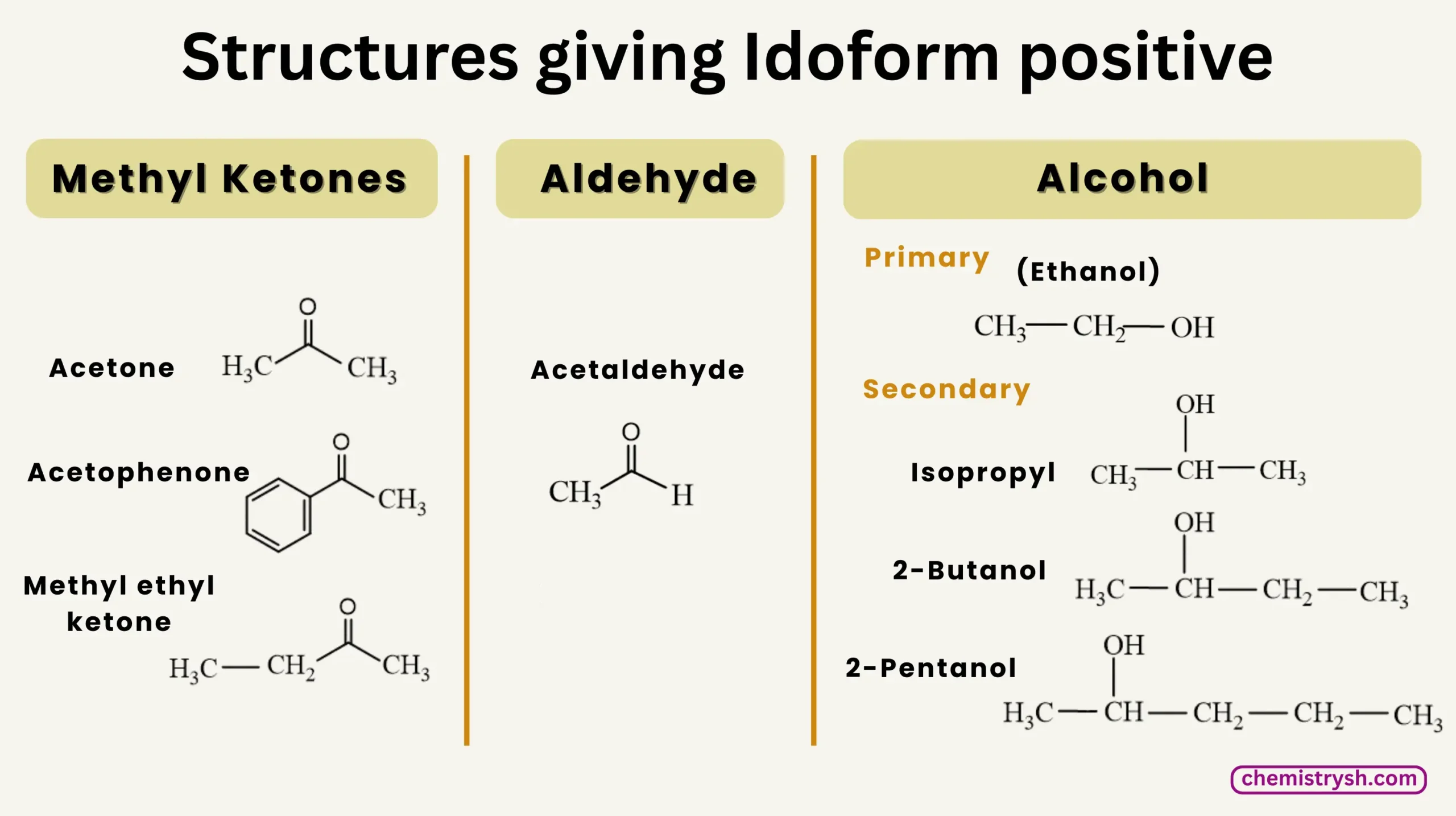
Note: Alcohols containing the –CH₃CH(OH)– group or those oxidizable to a methyl ketone (–CO–CH₃) give a positive iodoform test.
Iodoform Test Is Not Given By
- Methanol (CH₃OH): Oxidizes to formaldehyde (HCHO), not a methyl ketone
- Primary alcohols (except ethanol): 1-Propanol, 1-Butanol; oxidize to aldehydes/acids
- Tertiary alcohols: e.g., tert-Butanol; do not oxidize to carbonyl compounds
- Aldehydes (except acetaldehyde): Formaldehyde, benzaldehyde; lack –CO–CH₃
- Ketones other than methyl ketones: Cyclohexanone, benzophenone; no –CO–CH₃ group
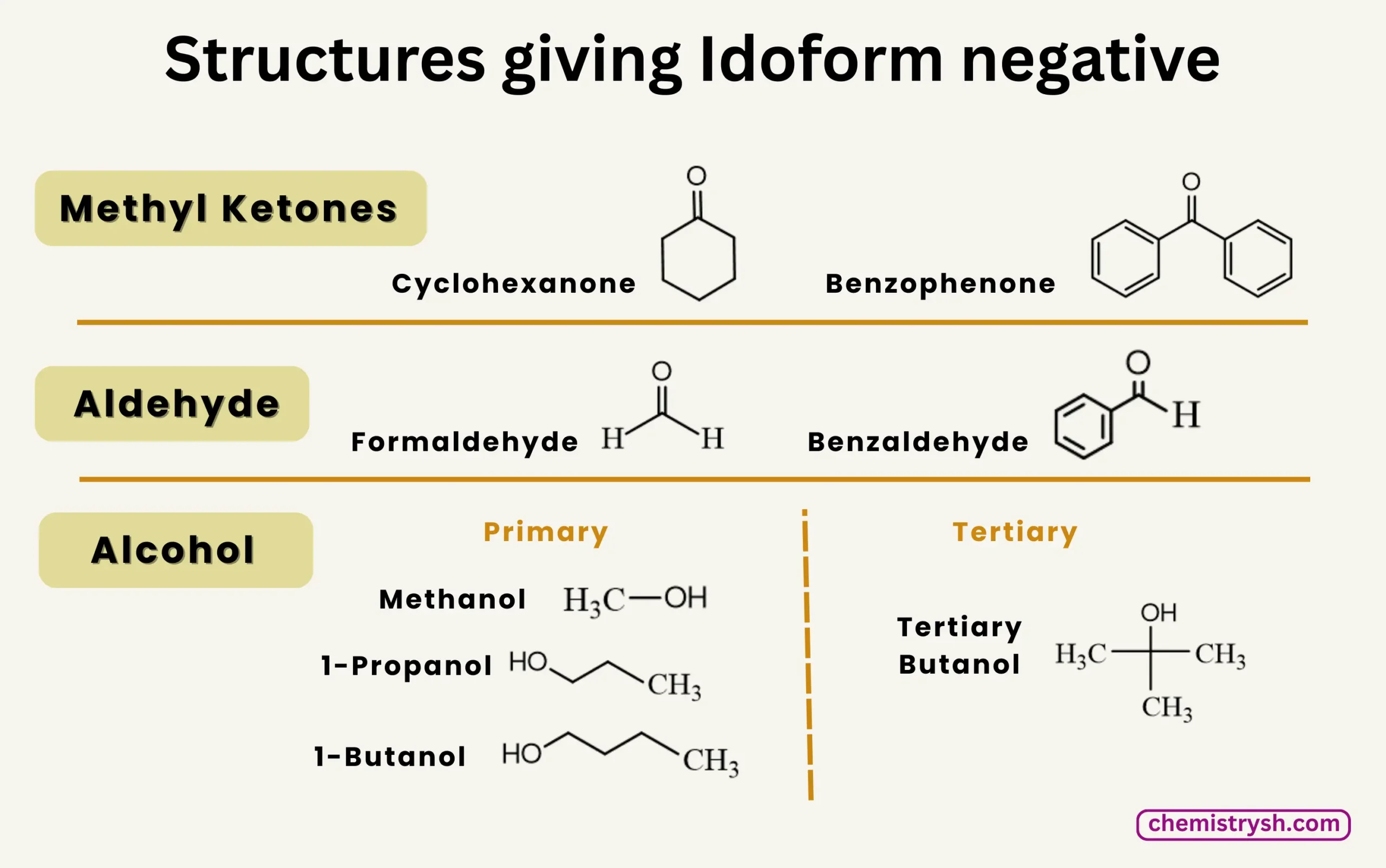
Note: Compounds without CH₃CO– or CH₃CH(OH)– do not give yellow iodoform crystals.
Iodoform Reaction
The iodoform reaction is used to detect methyl ketones or alcohols that can be oxidized to methyl ketones. Compounds containing the –CO–CH₃ or –CH₃CH(OH)– group react with I₂ in the presence of NaOH to form iodoform (CHI₃), a yellow crystalline solid with a characteristic medicinal odor.
This reaction serves as an important qualitative test and illustrates the principles of oxidation, halogenation, and carbon–carbon cleavage in organic chemistry.
Common compounds that give a positive iodoform reaction include acetone (CH₃COCH₃), ethanol (CH₃CH₂OH), acetaldehyde (CH₃CHO), 2-propanol (CH₃CHOHCH₃), and acetophenone (C₆H₅COCH₃).
The appearance of yellow CHI₃ crystals indicates that the compound is either one of these or a methyl ketone/secondary alcohol that can form a methyl ketone.
Why Is Iodoform Yellow?
Iodoform appears yellow because iodine atoms contain large, easily polarizable electron clouds. These heavy atoms influence electronic transitions in the molecule, causing absorption of blue light and reflection of yellow light.

Chemical Reactions of The Iodoform Test
When an organic compound reacts with iodine (I₂) in a basic solution (NaOH), it undergoes oxidative cleavage, producing a carboxylate salt and a yellow precipitate of iodoform (CHI₃) with a characteristic medicinal odor.
Example of some compounds giving iodoform reaction is given below:
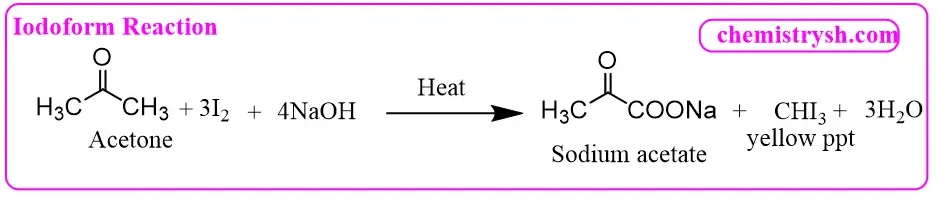
1. Acetone (CH₃COCH₃) – Methyl Ketone
Acetone reacts directly with iodine and NaOH to give iodoform and sodium acetate.
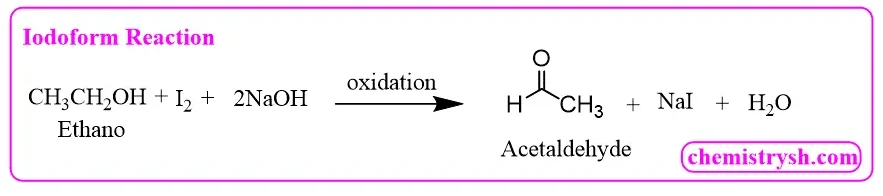
2. Ethanol (CH₃CH₂OH) – Primary Alcohol
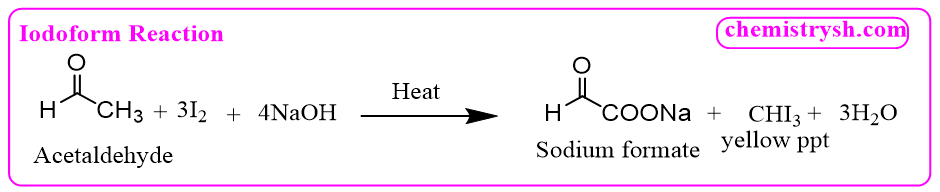
Ethanol is first oxidized to acetaldehyde, which then undergoes the iodoform reaction.
Step 1: Oxidation to acetaldehyde
Step 2: Iodoform reaction of acetaldehyde
3. Acetophenone (C₆H₅COCH₃) – Aromatic Methyl Ketone
Acetophenone reacts with iodine and NaOH to form iodoform and sodium benzoate.
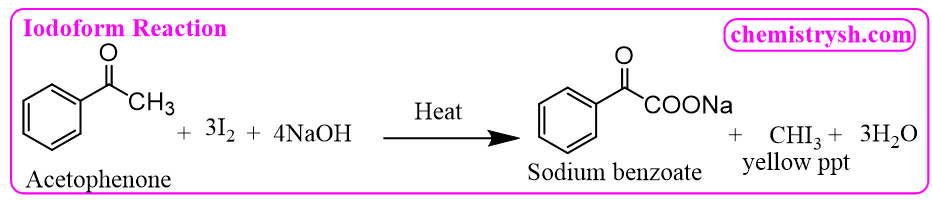
Note: The formation of yellow CHI₃ crystals indicates that the tested compound is a methyl ketone or an alcohol that can be oxidized to a methyl ketone.
Iodoform Reaction: Starting Compounds, Products, Carbon Changes, and CHI₃
|
Starting Compound (Formula) |
Final Carboxylate Salt (Formula) |
Iodoform Produced |
Carbon Change |
|
|---|---|---|---|---|
|
Acetone (CH₃COCH₃) |
Sodium Acetate (CH₃COONa) |
CHI₃ (s) |
Loses 1 Carbon |
|
|
Acetaldehyde (CH₃CHO) |
Sodium Formate (HCOONa) |
CHI₃ (s) |
Loses 1 Carbon |
|
|
Ethanol (CH₃CH₂OH) |
Sodium Formate (HCOONa) |
CHI₃ (s) |
Loses 1 Carbon |
|
|
2-Propanol (CH₃CH(OH)CH₃) |
Sodium Acetate (CH₃COONa) |
CHI₃ (s) |
Loses 1 Carbon |
|
|
2-Butanone (CH₃COCH₂CH₃) |
Sodium Propionate (CH₃CH₂COONa) |
CHI₃ (s) |
Loses 1 Carbon |
|
|
Sodium Propionate (CH₃CH₂COONa) |
CHI₃ (s) |
Loses 1 Carbon |
|
|
Acetophenone (C₆H₅COCH₃) |
Sodium Benzoate (C₆H₅COONa) |
CHI₃ (s) |
Loses 1 Carbon |

Note: Formation of iodoform CHI₃ (Triiodomethane), appears as yellow crystals with a distinctive antiseptic smell.
Mechanism of the Iodoform Reaction
- The reaction occurs in alkaline medium (NaOH) with iodine (I₂).
- Halogenation: OH acts as a base, removing the alpha-hydrogen from –CO–CH₃, allowing iodine substitution to form a tri-iodo intermediate (–CI₃).
- Cleavage: OH acts as a nucleophile, attacking the carbonyl carbon and facilitating C–C bond cleavage.
- Formation of iodoform (CHI₃): The tri-iodomethyl group leaves as CHI₃ (yellow crystals).
- Formation of carboxylate salt: The remaining carbonyl fragment becomes a carboxylate ion (R–COO⁻).
- The overall mechanism demonstrates halogenation, oxidation, and cleavage occurring sequentially.
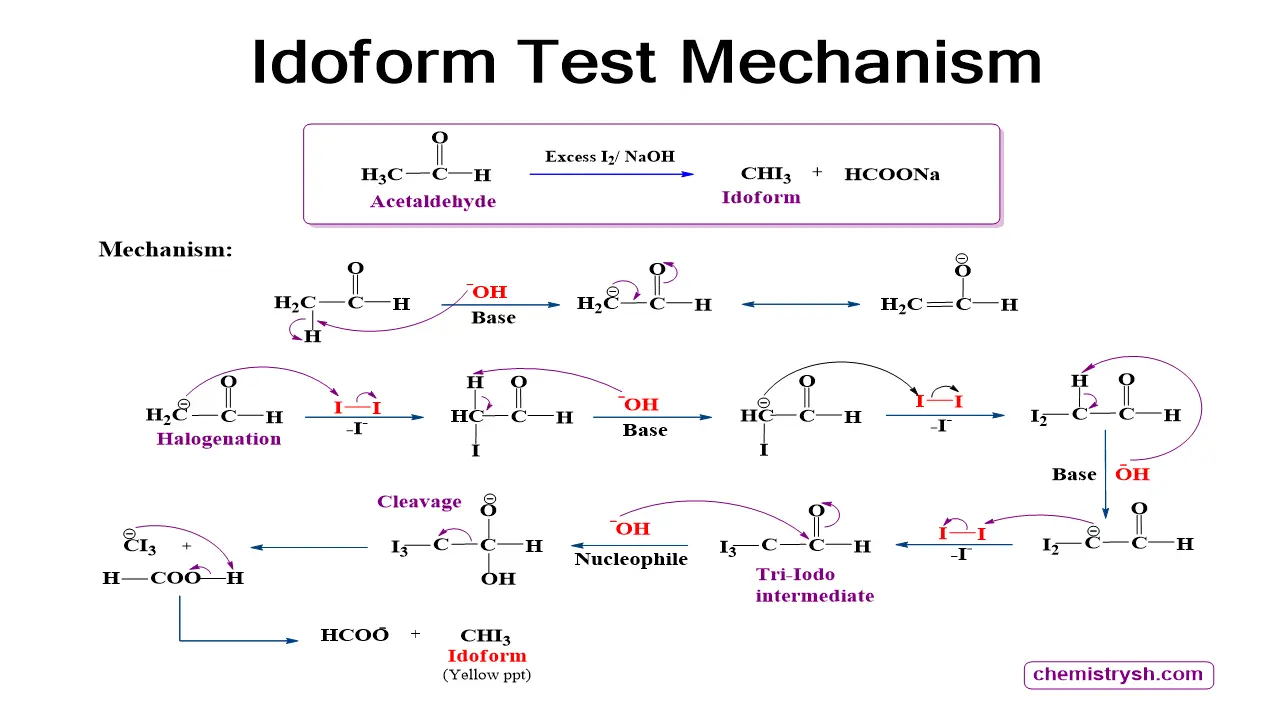
Role of Hydroxide ion (OH⁻) in Iodoform Reaction
Hydroxide Ion as a Base (OH⁻):
OH⁻ acts as a strong base by abstracting the acidic α-hydrogen of a methyl ketone, forming an enolate ion. This step is essential for successive α-halogenation of the methyl group
Hydroxide Ion as a Nucleophile (OH⁻):
OH⁻ also acts as a nucleophile, attacking the carbonyl carbon of the trihalomethyl ketone intermediate. This facilitates C–C bond cleavage, producing a carboxylate ion and the haloform (CHX₃).
Chemistry behind the Iodoform Reaction
- Structural Requirement: A compound must contain a methyl ketone (CH₃–CO–) or a secondary alcohol with CH₃–CH(OH)– that can be oxidized to a methyl ketone. Example: Ethanol (CH₃CH₂OH) → oxidized to acetaldehyde (CH₃CHO) → gives a positive test.
- Role of Iodine (I₂): Acts as a halogenating and oxidizing agent, introducing iodine at the alpha-carbon and oxidizing secondary alcohols to ketones.
- Role of Hydroxide Ions (OH⁻): As a base, abstracts alpha-hydrogens to form enolate ions for halogenation. As a nucleophile, attacks the carbonyl carbon, facilitating C–C bond cleavage, producing iodoform (CHI₃) and a carboxylate ion.
- Selectivity: The combination of structural requirements and this reaction mechanism explains why only certain compounds give a positive iodoform test.
Why Only Iodine Is Preferred in the Haloform Test?
Although haloform reactions can occur with chlorine or bromine, iodine is preferred because iodoform (CHI₃) forms a stable, bright yellow solid precipitate. Chloroform and bromoform remain liquids, making iodine the best choice for qualitative identification.
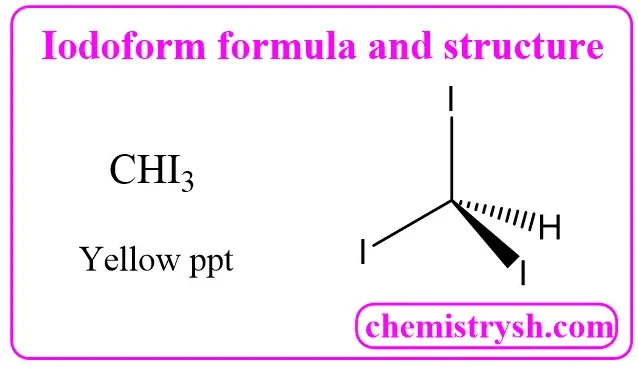
Iodoform (CHI₃) – Formula, Structure, Uses, and Identification
- Formula & Identity: Iodoform, also known as triiodomethane, has the chemical formula CHI₃.
- Structure: It features a central carbon atom bonded to three iodine atoms and one hydrogen in a tetrahedral geometry.
- Medicinal Use: Historically, iodoform was used as a potent antiseptic that slowly releases iodine to disinfect wounds and aid healing.
- Modern Applications: Today, iodoform is applied in dental care, particularly in antiseptic pastes and root canal filling materials.
- Laboratory Identification: Iodoform can be confirmed by the formation of its characteristic yellow crystals with the medicinal odor during tests.
Iodoform Test: Observation and Result Interpretation
- Observation: Yellow precipitate (CHI₃) + Antiseptic smell = Positive Test
- Negative test: No precipitate formation

Tabular Summary: Compounds Giving a Positive Iodoform Test and Their Products
|
Compound Name (Structure) |
Compound Type |
Products Formed |
|
|---|---|---|---|
|
Acetone |
Methyl ketone |
CHI₃ (Iodoform) + CH₃COONa (Sodium acetate) |
|
|
Methyl ketone |
CHI₃ (Iodoform) + C₂H₅COONa (Sodium propanoate) |
|
|
Acetophenone |
Methyl ketone |
CHI₃ (Iodoform) + C₆H₅COONa (Sodium benzoate) |
|
|
Ethanol |
Alcohol |
CHI₃ (Iodoform) + HCOONa (Sodium formate) |
|
|
Secondary alcohol |
CHI₃ (Iodoform) + CH₃COONa (Sodium acetate) |
|
|
2-Butanol |
Secondary alcohol |
CHI₃ (Iodoform) + C₂H₅COONa (Sodium propanoate) |
|
|
Acetaldehyde |
Aldehyde |
|
Comparison of Iodoform Reaction and Haloform Reaction:
|
Feature |
Iodoform Reaction |
|
|
|---|---|---|---|
|
Specific haloform reaction producing iodoform (CHI₃) |
General reaction producing haloforms (CHX₃) |
|
|
Halogen Used |
|
Chlorine, bromine, or iodine (Cl₂, Br₂, I₂) |
|
|
Yellow precipitate of iodoform (CHI₃) |
Chloroform (CHCl₃), bromoform (CHBr₃), or iodoform (CHI₃) |
|
|
Substrates |
Methyl ketones, ethanol, secondary alcohols with –CH₃CHOH– |
Same class of substrates |
|
|
Reaction Medium |
Alkaline (NaOH/KOH) |
Alkaline (NaOH/KOH) |
|
|
Purpose |
|
Both preparative and analytical |
|
|
Characteristic medicinal smell |
Depends on haloform formed |
|
|
Scope |
Narrow (iodine only) |
Broader (three halogens) |
Iodoform Test: Importance and Applications
- It provides a quick visual confirmation of specific functional groups, particularly methyl ketones and ethanol.
- The test is especially useful for:
- Distinguishing ethanol from other primary alcohols.
- Identifying methyl ketones among other carbonyl compounds.
- Historically, iodoform was applied as an antiseptic in medical treatments, though this use is now largely discontinued.
- The test demonstrates key organic chemistry concepts, including:
- Halogenation
- Enolate formation
- Nucleophilic substitution reactions
- In qualitative organic analysis, the iodoform test is often combined with other reactions to systematically identify unknown compounds and confirm the presence of specific functional groups.
Summary of the Iodoform Reaction
- The iodoform reaction is a qualitative test for methyl ketones and alcohols oxidizable to methyl ketones.
- Compounds with CH₃–CO– or CH₃–CH(OH)– groups react with iodine in a basic medium to produce yellow iodoform crystals (CHI₃) and a carboxylate ion.
- The hydroxide ion (OH⁻) acts both as a base, abstracting alpha-hydrogens, and as a nucleophile, attacking the carbonyl carbon.
- The reaction involves halogenation, oxidative cleavage of the C–C bond, and formation of the carboxylate, demonstrating key organic chemistry principles.
- The formation of the distinctive yellow precipitate provides a quick visual confirmation and is useful in laboratory identification and educational demonstrations.
Complementary Tests for Iodoform Test:
- 2,4-DNP Test: Confirms the presence of a carbonyl group (aldehyde or ketone).
- Tollen’s Test: Distinguishes acetaldehyde from methyl ketones; positive only for aldehydes.
- Benedict’s Test: Confirms aliphatic aldehydes such as acetaldehyde.
- Lucas Test: Identifies secondary alcohols that give a positive iodoform test.
- Oxidation Test (KMnO₄): Confirms secondary alcohols by oxidation to methyl ketones.
- Schiff’s Test: Confirms the presence of aldehydes.
- Sodium Bicarbonate Test – Rules out the presence of carboxylic acids by producing effervescence (CO₂ gas) if present
How to Differentiate Alcohol, Acetone, and Aldehyde When All Give a Positive Iodoform Test.
One of the most common challenges in the organic chemistry laboratory arises when different compounds give same test results, as in the case of the iodoform test, making identification more difficult.
A real case problem:
- Ethanol, acetone, and aldehydes are neutral organic compounds (checked by litmus paper test).
- All of the above give yellow precipitates of iodoform (CHI₃) check other coloured compounds.
- the iodoform test alone cannot be used to distinguish between ethanol, acetone, and aldehydes.
- To overcome this limitation, other chemical tests such as the 2,4-dinitrophenylhydrazine (2,4-DNPH) test, silver mirror (Tollen) test, and Fehling’s solution test are required for proper differentiation.
Chemical Tests for Identifying Ethanol, Acetaldehyde, and Acetone (2,4-DNPH, Tollens’, Fehling’s, Iodoform)
|
2,4-DNPH Test |
Silver Mirror Test |
Fehling’s Test |
Iodoform Test |
Conclusion |
|
|---|---|---|---|---|---|---|
|
Case 1 |
No reaction |
No reaction |
No reaction |
Positive (yellow precipitate) |
Ethanol |
|
|
Case 2 |
Positive (orange precipitate) |
Positive (silver mirror formed) |
Positive (brick-red precipitate) |
Positive (yellow precipitate) |
Acetaldehyde |
|
|
Case 3 |
Positive (orange precipitate) |
No reaction |
No reaction |
Positive (yellow precipitate) |
Acetone |
Viva questions
1. Is iodoform a name reaction?
Yes, the iodoform reaction is considered a name reaction in organic chemistry. It is a specific type of haloform reaction and is named after the characteristic product formed – iodoform (CHI₃). The reaction is also classified under haloform reactions, which include chloroform and bromoform reactions. Name reactions are well-known, commonly used reactions that are often named after their discoverers or characteristic products.
2. Who can give an iodoform reaction?
The following compounds can give an iodoform reaction:
- Compounds with CH₃CO- group (methyl ketones): Acetone, 2-butanone, acetophenone, etc.
- Compounds with CH₃CHOH- group (methyl carbinols): 2-propanol, 2-butanol, 2-pentanol, etc.
- Special cases: Ethanol (CH₃CH₂OH) – the only primary alcohol and Acetaldehyde (CH₃CHO) – the only aldehyde.
Requirement: The compound must have a methyl group directly attached to a carbonyl carbon OR a carbon bearing a hydroxyl group (which can be oxidized to a carbonyl).
3. Can isopropyl alcohol give a positive iodoform test?
Yes, isopropyl alcohol (2-propanol, CH₃CHOHCH₃) gives a positive iodoform test.
Reason: Isopropyl alcohol has the structure CH₃CHOH-CH₃, which contains the methyl carbinol group (CH₃CHOH-). During the reaction, it is oxidized to acetone (CH₃COCH₃), which then reacts to form the yellow iodoform precipitate.
Reaction: CH₃CHOHCH₃ → CH₃COCH₃ → CHI₃ (yellow precipitate)
4. Will propanol give an iodoform reaction?
It depends on which propanol:
- 1-Propanol (n-propanol, CH₃CH₂CH₂OH): NO – Does not give a positive iodoform test because it lacks the required CH₃CO- or CH₃CHOH- structure.
- 2-Propanol (isopropanol, CH₃CHOHCH₃): YES – Gives a positive iodoform test because it has the CH₃CHOH- structure.
Key point: Only 2-propanol (isopropyl alcohol) gives the iodoform reaction, not 1-propanol.
5. What is the iodoform reaction of Butan-2-ol?
Butan-2-ol (CH₃CH₂CHOHCH₃ or CH₃CHOH-CH₂CH₃) gives a positive iodoform test.
Mechanism:
- Oxidation: Butan-2-ol is oxidized to butan-2-one (methyl ethyl ketone): CH₃CH₂CHOHCH₃ + I₂ + NaOH → CH₃CH₂COCH₃ + NaI + H₂O
- Halogenation: The methyl group of the ketone undergoes sequential iodination: CH₃CH₂COCH₃ + 3I₂ + 3NaOH → CH₃CH₂COCI₃ + 3NaI + 3H₂O
- Cleavage: Base-catalyzed cleavage produces iodoform: CH₃CH₂COCI₃ + NaOH → CHI₃↓ (yellow precipitate) + CH₃CH₂COONa
Overall reaction: CH₃CH₂CHOHCH₃ + 4I₂ + 6NaOH → CHI₃ + CH₃CH₂COONa + 5NaI + 5H₂O
6. Can aldehyde give an iodoform reaction?
Only one aldehyde gives the iodoform reaction – Acetaldehyde (CH₃CHO).
Why acetaldehyde is positive:
- It has the required CH₃CO- structure
- The methyl group is adjacent to the carbonyl group
Why other aldehydes are negative:
- Formaldehyde (HCHO) – no methyl group
- Propionaldehyde (CH₃CH₂CHO) – methyl group not directly attached to carbonyl
- Benzaldehyde (C₆H₅CHO) – no methyl group attached to carbonyl
Acetaldehyde reaction: CH₃CHO + 3I₂ + 4NaOH → CHI₃ + HCOONa + 3NaI + 3H₂O
7. Can acetone give an iodoform reaction?
Yes, acetone gives a positive iodoform test. Acetone is actually one of the most common examples used to demonstrate this reaction.
Structure: CH₃COCH₃ (contains two CH₃CO- groups)
Reaction: CH₃COCH₃ + 3I₂ + 4NaOH → CHI₃↓ (yellow precipitate) + CH₃COONa + 3NaI + 3H₂O
Result: Formation of bright yellow precipitate of iodoform with characteristic antiseptic smell.
Acetone is often used in laboratories as a standard positive control for the iodoform test.
8. Does 2-pentanone give a positive iodoform test?
Yes, 2-pentanone (CH₃COCH₂CH₂CH₃) gives a positive iodoform test.
Reason: 2-pentanone is a methyl ketone with the structure CH₃CO-R (where R = CH₂CH₂CH₃). It contains the required CH₃CO- group.
Reaction: CH₃COCH₂CH₂CH₃ + 3I₂ + 4NaOH → CHI₃↓ + CH₃CH₂CH₂COONa + 3NaI + 3H₂O
Products: Yellow iodoform precipitate and sodium butyrate.
9. Can 3-pentanone give an iodoform reaction?
No, 3-pentanone (CH₃CH₂COCH₂CH₃) does NOT give a positive iodoform test.
Reason: 3-pentanone does not have a methyl group directly attached to the carbonyl carbon. Instead, it has ethyl groups (CH₂CH₃) on both sides of the carbonyl. The iodoform test specifically requires a CH₃CO- structure.
Key difference:
- 2-pentanone: CH₃CO-CH₂CH₂CH₃ ✓ (positive – has CH₃CO-)
- 3-pentanone: CH₃CH₂-CO-CH₂CH₃ ✗ (negative – no CH₃CO-)
10. Which will give a positive iodoform reaction?
Compounds that give a positive iodoform reaction include:
Methyl ketones:
- Acetone (CH₃COCH₃)
- 2-Butanone (CH₃COCH₂CH₃)
- 2-Pentanone (CH₃COCH₂CH₂CH₃)
- Acetophenone (CH₃COC₆H₅)
Secondary alcohols with CH₃CHOH- structure:
- 2-Propanol (CH₃CHOHCH₃)
- 2-Butanol (CH₃CHOHCH₂CH₃)
- 2-Pentanol (CH₃CHOHCH₂CH₂CH₃)
Special cases:
- Ethanol (CH₃CH₂OH) – only primary alcohol
- Acetaldehyde (CH₃CHO) – only aldehyde
General rule: Any compound with CH₃CO- or CH₃CHOH- structure, or that can be oxidized to these structures.
11. What happens when iodine reacts with NaOH?
When iodine reacts with sodium hydroxide, a disproportionation reaction occurs:
Reaction: I₂ + 2NaOH → NaI + NaOI + H₂O
Or in ionic form: I₂ + 2OH⁻ → I⁻ + OI⁻ + H₂O
Products formed:
- Sodium iodide (NaI) – iodine is reduced (I₂ → I⁻)
- Sodium hypoiodite (NaOI) – iodine is oxidized (I₂ → OI⁻)
- Water (H₂O)
In the iodoform test: The sodium hypoiodite (NaOI) acts as both an oxidizing agent and a source of electrophilic iodine (I⁺), which is necessary for:
- Oxidizing alcohols to ketones or aldehydes
- Halogenating the methyl group adjacent to the carbonyl
This is why NaOH is essential in the iodoform reaction – it generates the reactive iodine species needed for the reaction.
12. Which alcohols (primary, secondary, or tertiary) give a positive iodoform test?
PRIMARY ALCOHOLS:
- Only ethanol (CH₃CH₂OH) gives positive test
- All other primary alcohols (methanol, 1-propanol, 1-butanol, etc.) give negative test
SECONDARY ALCOHOLS:
- Only those with CH₃CHOH- structure give positive test
- Examples: 2-propanol, 2-butanol, 2-pentanol
- Secondary alcohols without methyl group adjacent to -OH give negative test
- Example: 3-pentanol (CH₃CH₂CHOHCH₂CH₃) is negative
TERTIARY ALCOHOLS:
- All tertiary alcohols give negative test
- Examples: tert-butanol, 2-methyl-2-propanol
- Reason: Cannot be easily oxidized to ketones
Summary:
- Primary: Only ethanol (positive)
- Secondary: Only those with CH₃CHOH- (positive)
- Tertiary: None (all negative)
13. What is the mechanism of the iodoform reaction?
The iodoform reaction proceeds through the following mechanism:
Step 1: Oxidation (for alcohols)
If the starting material is an alcohol (CH₃CHOH-R), it is first oxidized to a methyl ketone: CH₃CHOH-R + I₂ + 2OH⁻ → CH₃CO-R + 2I⁻ + 2H₂O
Step 2: Enolization
The methyl ketone forms an enolate ion in basic solution: CH₃CO-R + OH⁻ ⇌ CH₂=C(O⁻)-R + H₂O
Step 3: Sequential Halogenation
The enolate ion reacts with iodine, and this process repeats three times:
First iodination: CH₂=C(O⁻)-R + I₂ → CH₂ICO-R + I⁻
Second iodination: CH₂ICO-R + OH⁻ → CHI=C(O⁻)-R + H₂O CHI=C(O⁻)-R + I₂ → CHI₂CO-R + I⁻
Third iodination: CHI₂CO-R + OH⁻ → CI₂=C(O⁻)-R + H₂O CI₂=C(O⁻)-R + I₂ → CI₃CO-R + I⁻
Step 4: Nucleophilic Acyl Substitution (Cleavage)
The tri-iodo compound undergoes base-catalyzed cleavage: CI₃CO-R + OH⁻ → CI₃⁻ + R-COO⁻
Step 5: Protonation
The triiodomethyl anion (CI₃⁻) is immediately protonated to form iodoform: CI₃⁻ + H₂O → CHI₃ + OH⁻
Overall: The reaction involves oxidation, multiple halogenations, and cleavage to produce the characteristic yellow iodoform precipitate.
Why CI₃⁻ is a good leaving group: The three iodine atoms stabilize the negative charge through their electron-withdrawing effect, making CI₃⁻ a relatively stable leaving group.
14. What is the iodoform test used to distinguish between?
The iodoform test is used to distinguish between:
1. Alcohols:
- Ethanol vs. other primary alcohols (methanol, 1-propanol, 1-butanol)
- 2-Propanol vs. 1-propanol
- Secondary alcohols with CH₃CHOH- vs. those without
- 2-Butanol (positive) vs. 1-butanol (negative)
2. Ketones:
- Methyl ketones vs. non-methyl ketones
- Acetone vs. diethyl ketone
- 2-Pentanone (positive) vs. 3-pentanone (negative)
- Acetophenone (positive) vs. benzophenone (negative)
3. Aldehydes:
- Acetaldehyde vs. all other aldehydes
- Acetaldehyde (positive) vs. formaldehyde, propionaldehyde, benzaldehyde (negative)
4. General Organic Compounds:
- Compounds with CH₃CO- or CH₃CHOH- groups vs. those without
- Primary, secondary, and tertiary alcohols from each other
- Aliphatic vs. aromatic ketones (when aromatic ketones lack the CH₃CO- group)
5. Structural Isomers:
- Butan-2-ol vs. butan-1-ol
- 2-Butanone vs. butanal
- Pentane-2-one vs. pentane-3-one
Viva Questions
FAQ’s
Multiple Choice Questions
MCQ 1
1. Which type of ketone gives a positive iodoform test?
A. Aromatic Ketone
B. Methyl Ketone
C. Cyclic Ketone
D. Aldehyde
MCQ 2
2. The yellow precipitate formed in the iodoform test is:
A. CHCl3
B. CBr3H
C. CHI3
D. C2H5I
MCQ 3
3. Which aldehyde gives a positive iodoform test?
A. Acetaldehyde
B. Formaldehyde
C. Benzaldehyde
D. Propanal
MCQ 4
4. What is the role of iodine in the iodoform reaction?
A. Acts as a solvent
B. Acts as halogenating and oxidizing agent
C. Neutralize the base
D. Provides the precipitates
MCQ 5
5. Which alcohol gives a positive iodoform test?
A. Methanol
B. 1-Propanol
C. 1-Butanol
D. Ethanol
References
- Finar, I. L. (2012). Organic chemistry, Volume 1: Fundamental principles (6th ed., pp. 385-388). Pearson Education.
- International Union of Pure and Applied Chemistry. Compendium of chemical terminology (Gold Book).
- Lieber, E., & Levering, D. R. (1959). The iodoform test. Journal of Chemical Education, 36(11), 572.
- McMurry, J. (2015). Organic chemistry (9th ed., pp. 856-858). Cengage Learning.
- Morrison, R. T., & Boyd, R. N. (2010). Organic chemistry (7th ed., pp. 742-745). Pearson Education.







I got this site from my pal who informed me about this website and at
the moment this time I am visiting this website and reading very informative posts at this place.