Color of Organic Compounds:
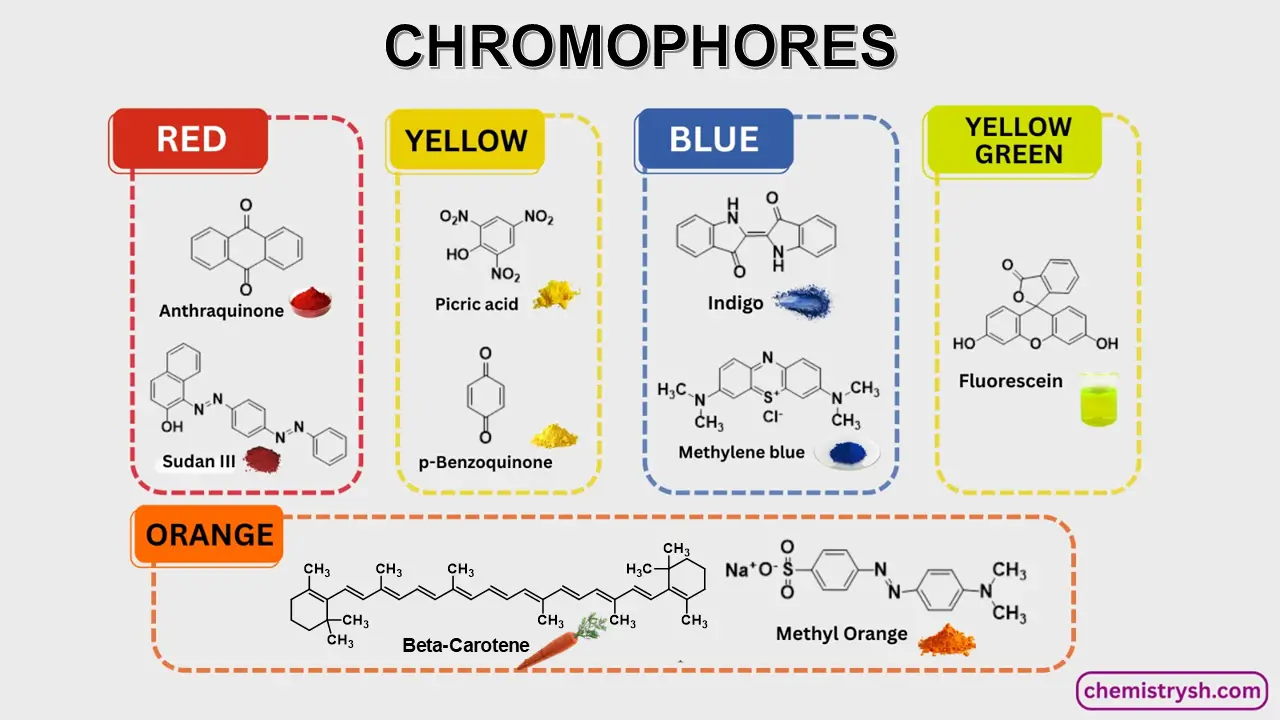
How the Color of Organic Compounds Helps in Organic Identification? Color is an important physical property in the preliminary identification of organic compounds. Compounds appear colored when they contain chromophores that absorb visible light, while colorless compounds lack these units. Thus, the presence of color mainly arises from chromophores within the molecular structure.
Phenolic and aromatic compounds also show distinctive color behavior when treated with specific reagents. Tests like the ferric chloride test use color development to confirm the presence of phenolic functional groups.
Why Organic Compounds are Colored?
Chromophores and Conjugation: The Engines of Color
A chromophore is a structural feature that absorbs light within the visible spectrum. Common chromophores include:
Color is influenced by conjugation, where overlapping p-orbitals allow electron delocalization, lowering the energy gap (ΔE) and shifting absorption into the visible region. Auxochromes (e.g., –OH, –NH₂) can further enhance or shift the color by modifying the electronic environment.
This principle of electronic transitions is also reflected in oxidation based reactions. In the Baeyer’s test, intense color changes occur due to shifts in oxidation state, clearly demonstrating how electronic structure controls the formation of colored species.
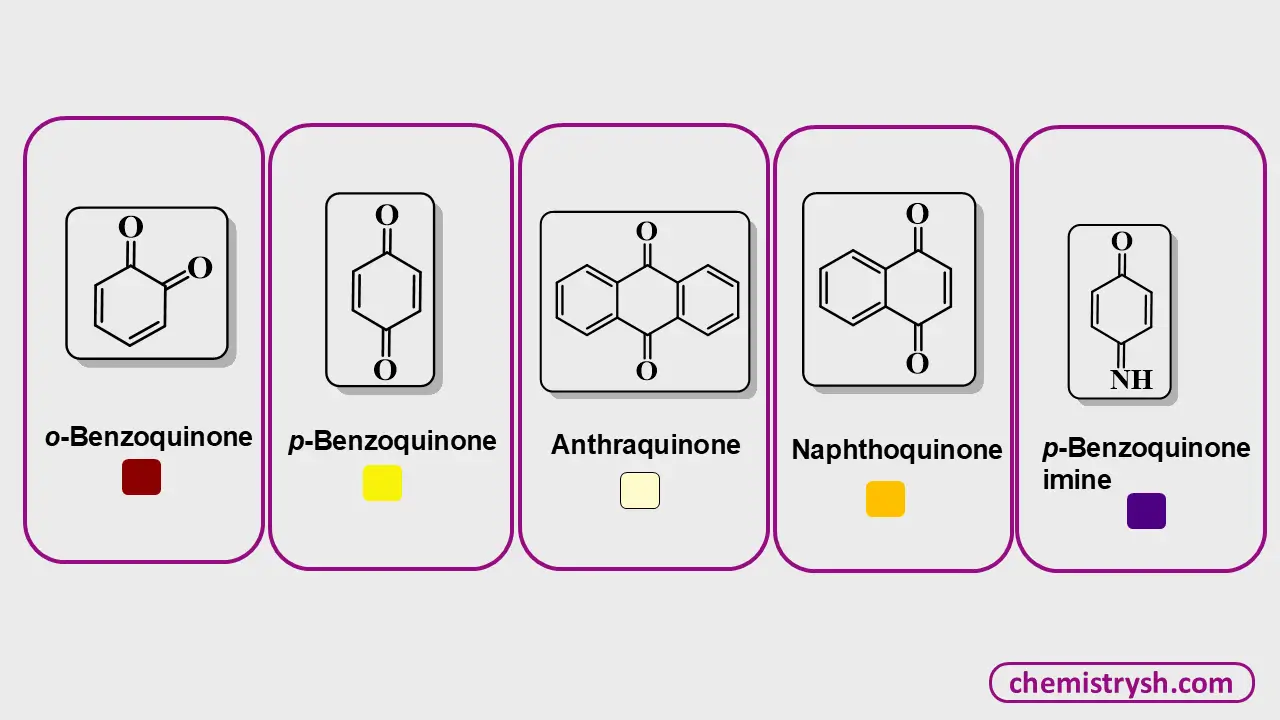
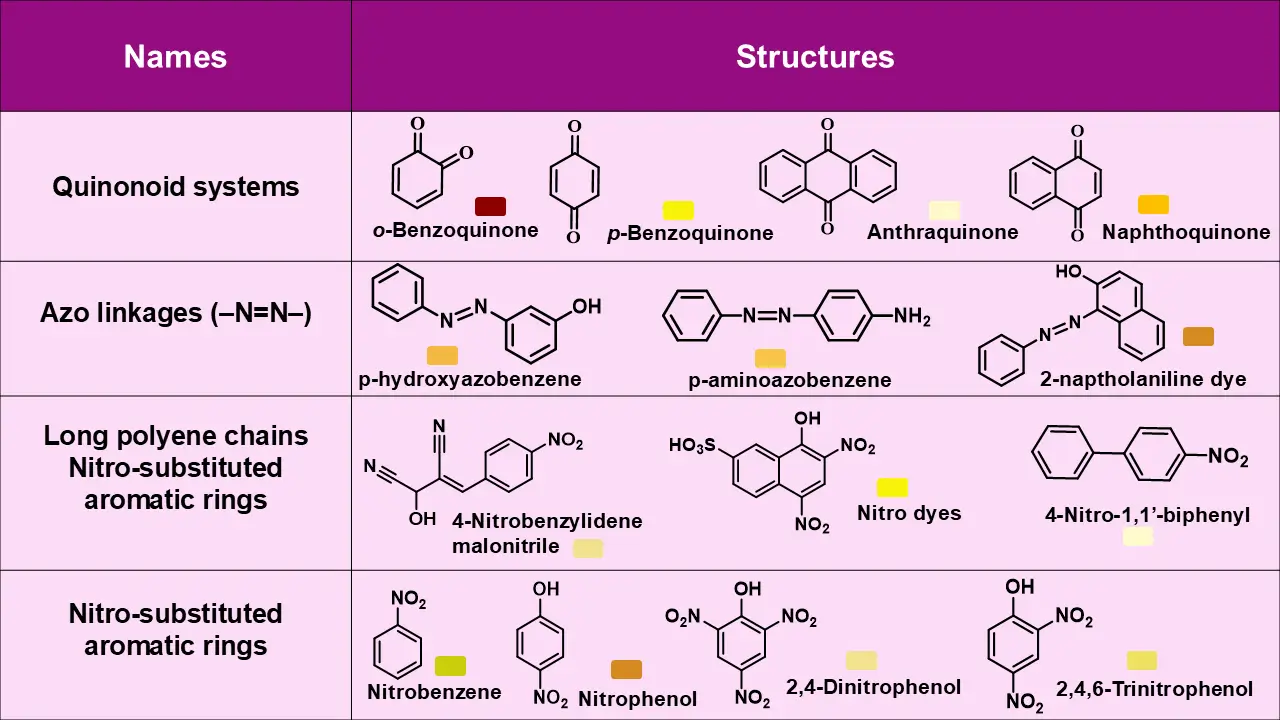
1. Quinones
Quinones are compounds containing a cyclohexadienedione ring system. For example, p-benzoquinone can be represented as:
The conjugation between the carbonyl groups and the ring double bonds produces a yellow color. Fused quinones, such as naphthoquinone, extend this conjugation, resulting in orange hues. Quinoid systems are consistently colored, and the color intensity increases with the size of the conjugated system.
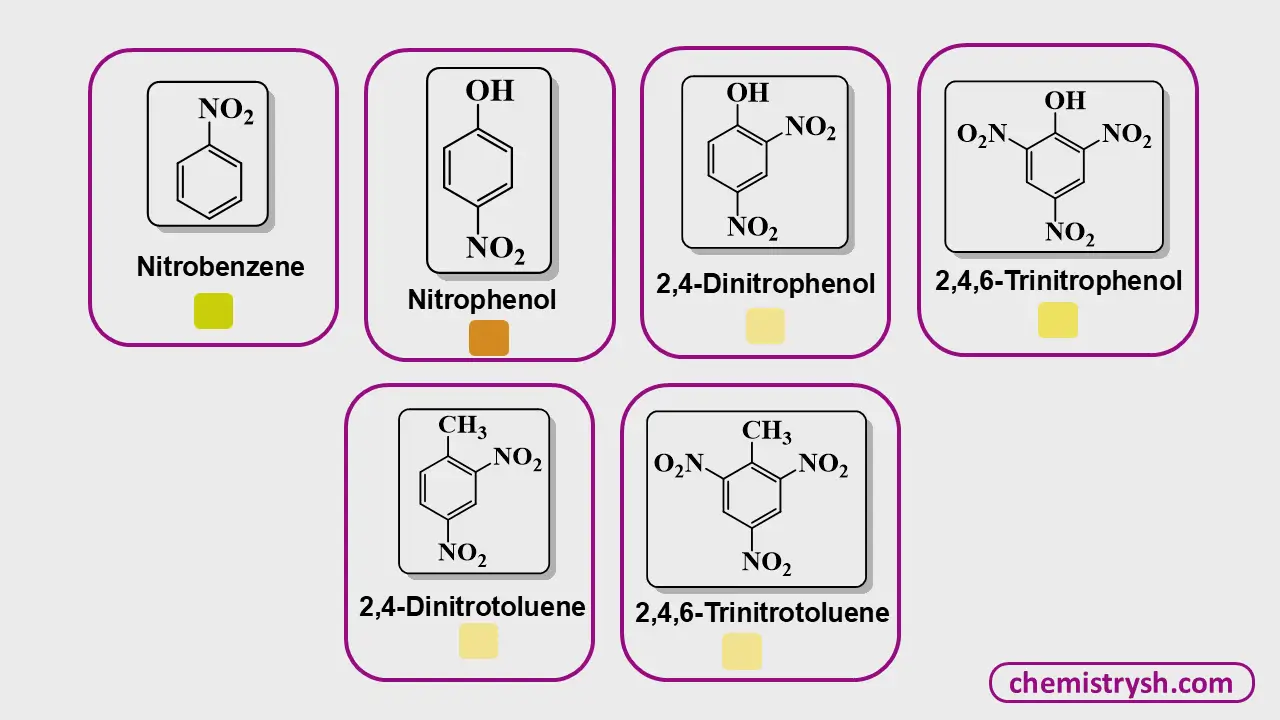
2. Nitro Aromatics
The introduction of nitro groups into aromatic systems produces color. For example:
The –NO₂ groups act as chromophores, and additional nitro groups increase conjugation, enhancing visible absorption
3. Azo Compounds
Azo compounds are formed via diazotization followed by coupling with aromatic compounds. Their general structure: Ar—N=N—Ar′
This linkage, combined with conjugated aromatic rings, produces bright colors ranging from yellow to red. Auxochromes on the aromatic rings, such as –OH or –NH₂, further intensify these colors. Azo dyes are a primary example of functional color in organic compounds.
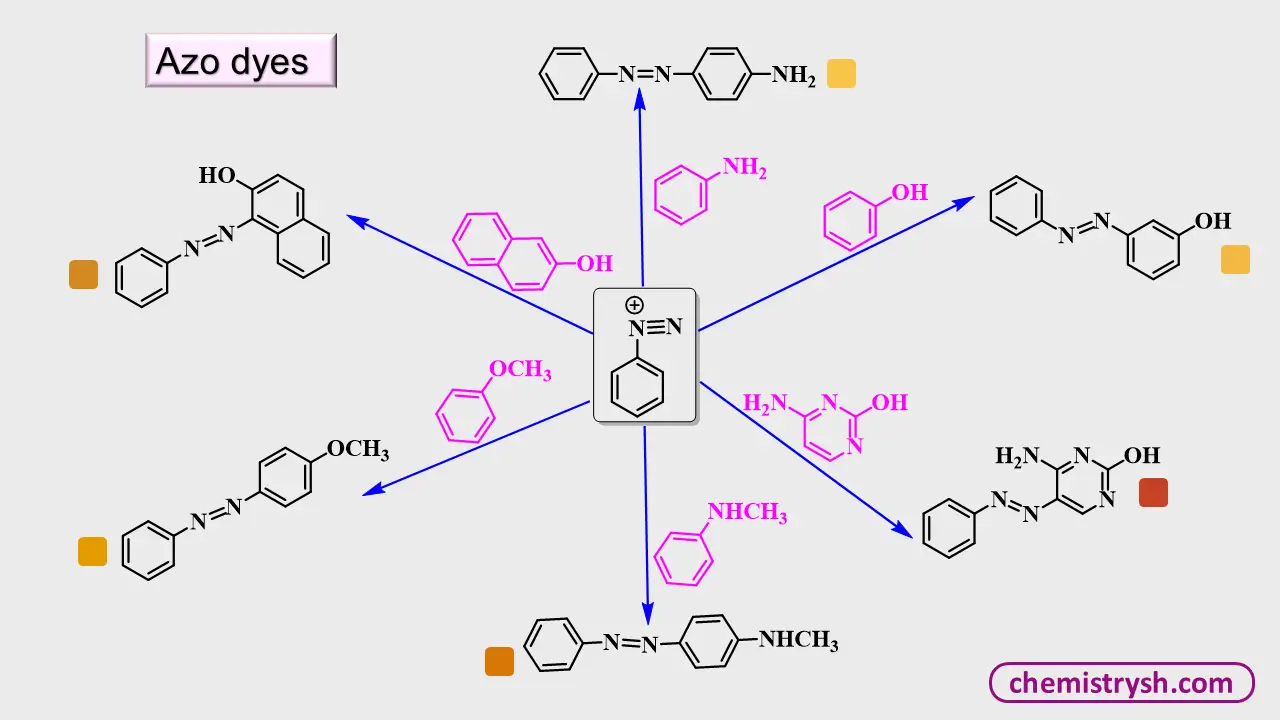
Did You Know
Azo dyes account for over 60% of synthetic dyes.
Diazotization reactions provide versatile intermediates for functionalized aromatic compounds.
Color can indicate structural features, making it an important tool in organic chemistry.
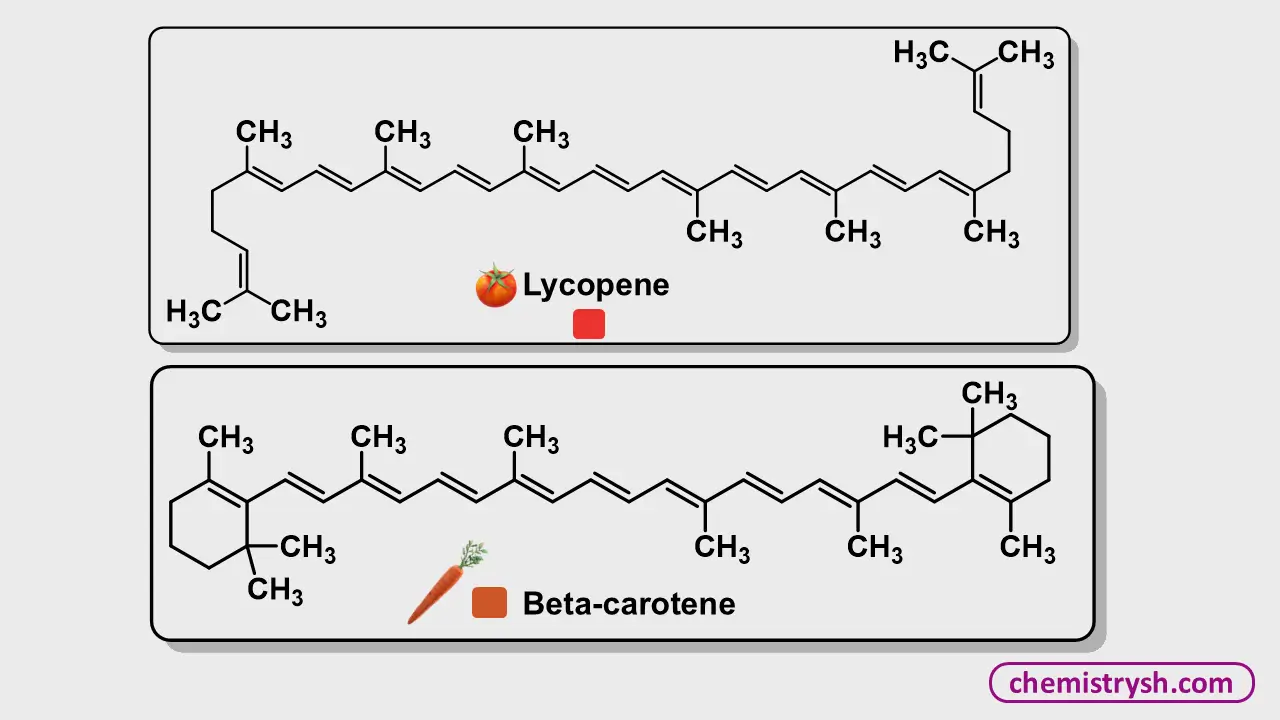
4. Polyenes
Linear conjugated double bonds (–C=C–)ₙ also absorb visible light. Short chains are colorless, but longer chains such as in β-carotene or lycopene appear orange or red:
The longer the conjugated chain, the lower the ΔE, and the more intense and red-shifted the color becomes.
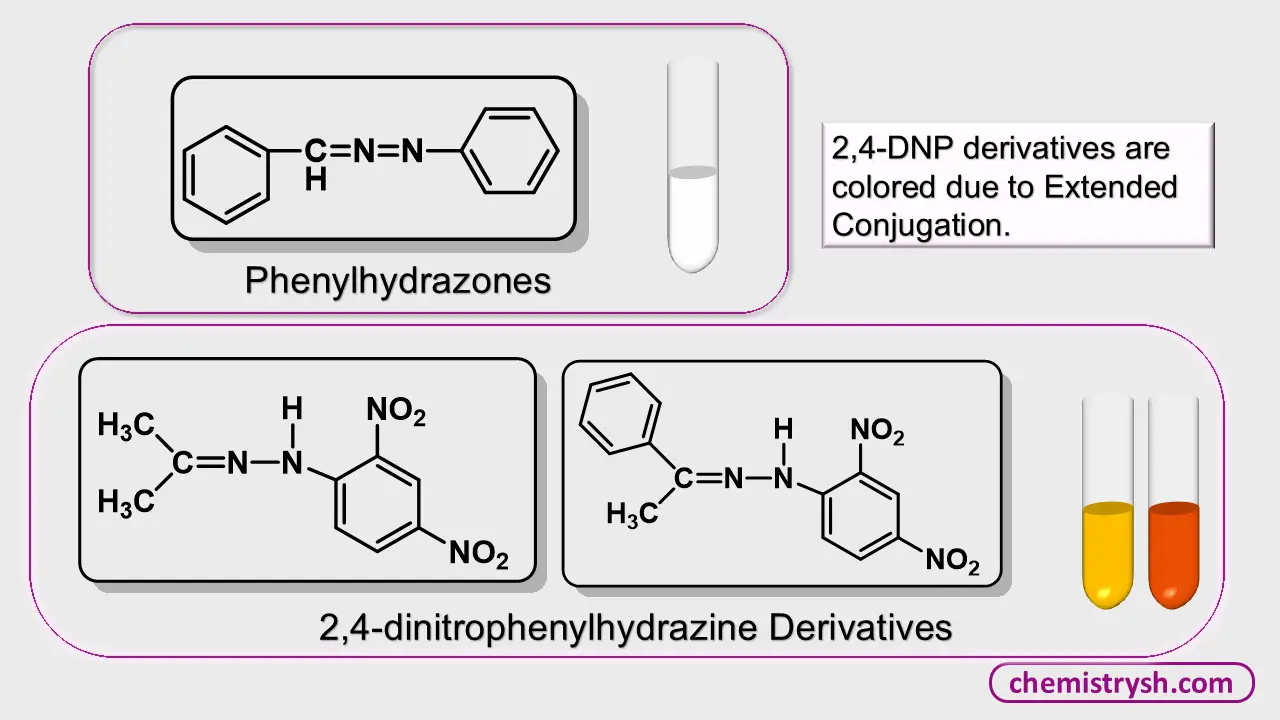
5. Hydrazones and Schiff Bases
Carbonyl compounds form colored derivatives with 2,4-dinitrophenylhydrazine (yellow/orange) while phenylhydrazones are usually white. For example:
The nitro groups act as chromophores, extending conjugation through the hydrazone linkage, and producing visible color. This principle underlies the widely used 2,4-DNPH test for carbonyl compounds.
Examples of Colored Organic Compounds
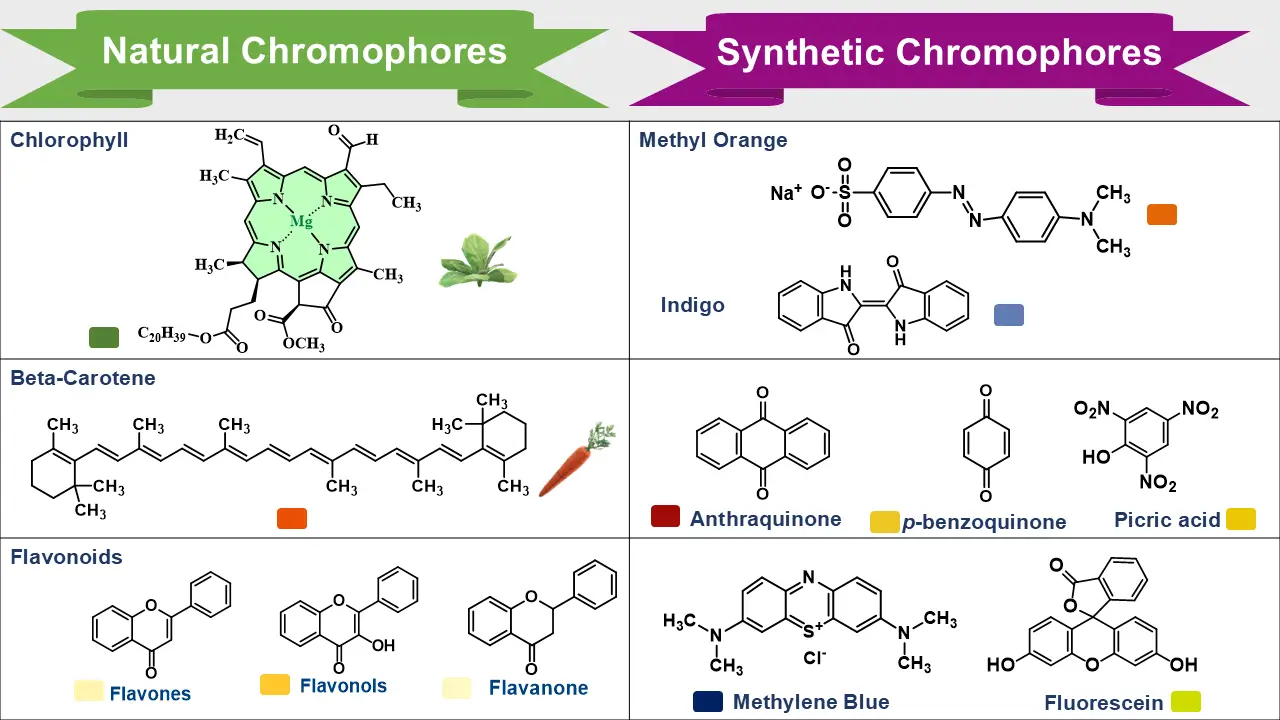
1. Natural Colored Compounds
- Carotenoids: Found in carrots and tomatoes; orange to red.
- Anthocyanins: Found in berries and flowers; red, purple, or blue depending on pH.
- Chlorophylls: Found in plants; green due to conjugated porphyrin rings.
2. Synthetic Colored Compounds
- Azo Dyes: Contain -N=N- group; widely used in textiles.
- Indigo: Classic blue dye for denim.
- Alizarin: Red dye derived from anthraquinone.
Applications of Colored Compounds
- Textile and Dye Industry – preparation of stable and bright azo dyes.
- Analytical Chemistry – detection of functional groups by observing color.
- Pharmaceutical Industry – dyes for drugs and pH indicators.
- Food Industry – natural and synthetic colorants.
- Biochemistry – study of pigments in plants and animals.
- Industrial Chemistry – synthesis of dyes, pigments, and chemical markers.
Conclusion
Conclusion: Logical Patterns for Students
The study of colored organic compounds reveals a logical and predictable relationship between structure and color:
- Chromophores absorb visible light.
- Extended conjugation lowers ΔE, shifting absorption into the visible spectrum.
- Auxochromes modify the intensity and wavelength of absorption.
· Structural features responsible for color include:
- Quinonoid systems
- Azo linkages (–N=N–)
- Long polyene chains
- Nitro-substituted aromatic rings
· Colored compounds can often be predicted by identifying these structural templates.
· Practical identification involves:
- Observing the physical color of the compound
- Performing functional group tests
- Analyzing the extent of structural conjugation
· Combined analysis helps students to predict or explain the color of unknown organic compounds effectively.
Viva questions
- What causes color in organic compounds?
- What is a chromophore?
- Why are azo compounds strongly colored?
- Give two examples of natural pigment s
- What is the role of auxochromes in color?
- Why is diazotization important in color formation?
Multiple Choice Questions
MCQ 1
1. Color in organic compounds is mainly due to:
A. Density
B. Chromophores
C. Solubility
D. Molecular mass
MCQ 2
2. Which group is responsible for azo dye color?
A. –OH
B. –NH₂
C. –N=N–
D. –COOH
MCQ 3
3. Chlorophyll is a:
A. Synthetic dye
B. Natural pigment
C. Indicator
D. Salt
MCQ 4
4. Greater conjugation results in
A. Color loss
B.Deeper color
C. Colorless compound
D. Decomposition
MCQ 5
5. Which of these is an auxochrome?
A. –NO₂
B. –OH
C. –N=N–
D. –C≡N
MCQ 6
6. Diazonium salts in diazotization are formed from
A. Secondary amines
B. Primary aromatic amines
C. Aliphatic amines
D. Alcohols
FAQ’s
References
- Brady, O. L. (1931). A simple qualitative test for aldehydes and ketones. Journal of the Chemical Society.
- Furniss, B. S., et al. Vogel’s Textbook of Practical Organic Chemistry.
- Reusch, W. (n.d.). Carbonyl compound identification. MCC Organic Chemistry.