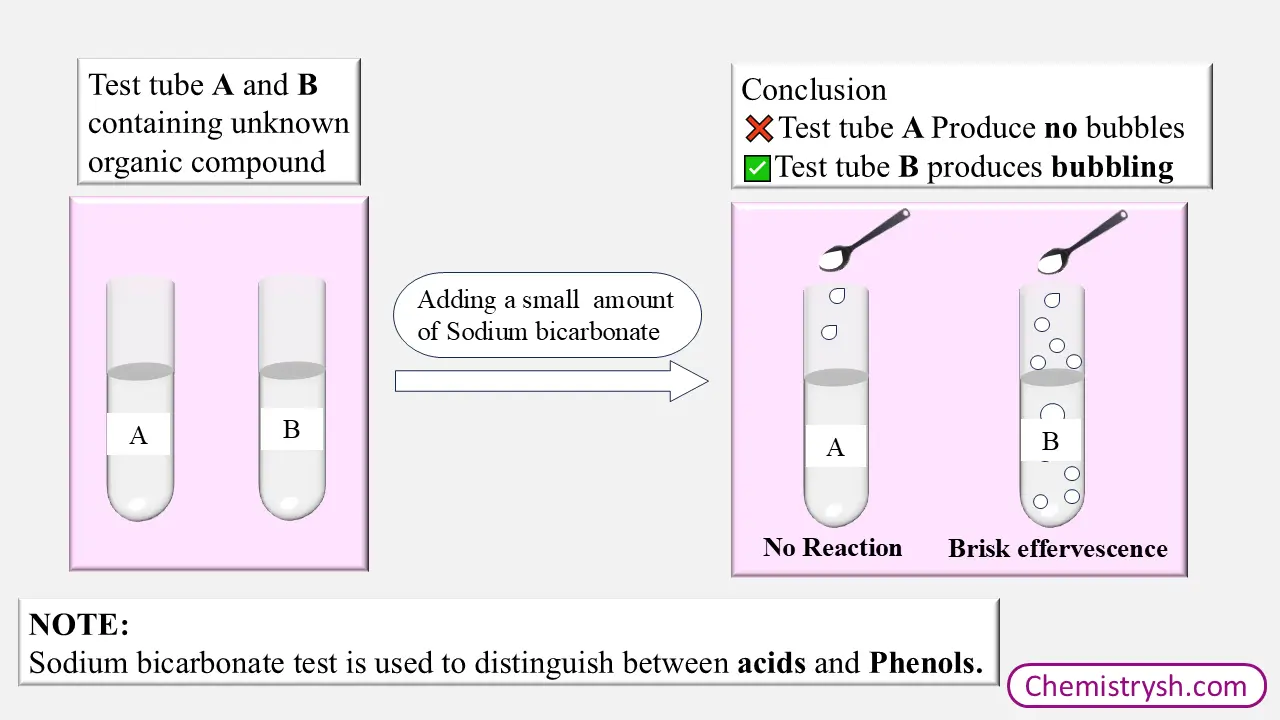
Introduction
The sodium bicarbonate test is a widely used laboratory test for the identification of carboxylic acids. It is based on the reaction of acidic compounds with aqueous sodium bicarbonate, which leads to the liberation of carbon dioxide gas (effervescence). This test is particularly useful for distinguishing carboxylic acids from phenols due to the difference in their acidic strength.
Carboxylic acids have low pKa values (≈ 4–5), indicating that they are relatively strong acids and can readily donate a proton to sodium bicarbonate, resulting in visible CO₂ evolution. In contrast, phenols possess higher pKa values (≈ 9–10) and are much weaker acids; therefore, they do not react with sodium bicarbonate under normal laboratory conditions. This difference in pKa values explains why carboxylic acids give a positive sodium bicarbonate test, while phenols give a negative test.
If the Compound is soluble in NaOH but it does not react with the sodium bicarbonate (NaHCO₃) then it is Phenol.
Did you know?
Carbon dioxide evolved in this test turns lime water milky, confirming the identity.
Sodium bicarbonate is a mild base, which makes this test selective for stronger organic acids.
What is the Sodium Bicarbonate Test?
The sodium bicarbonate test is a qualitative chemical test used to detect the presence of the carboxylic acid functional group (–COOH).
In this test, the organic compound is treated with sodium bicarbonate solution (NaHCO₃). The formation of effervescence confirms the presence of carboxylic acids.
Principle of Sodium Bicarbonate Test
The test is based on an acid–base reaction. Carboxylic acids are stronger acids than carbonic acid.
When a carboxylic acid reacts with sodium bicarbonate, it displaces carbonic acid, which decomposes to release carbon dioxide gas (CO₂). Phenols are weaker acids and cannot displace carbonic acid.
Carboxylic acids react with sodium bicarbonate to form sodium carboxylate, water, and carbon dioxide gas. The liberation of carbon dioxide results in brisk effervescence, which is the key positive observation of the sodium bicarbonate test.
Chemical equation:
RCOOH + NaHCO3 → RCOONa + H2O + CO2↑
The evolution of CO₂ gas confirms the presence of a carboxylic acid group, as only acids strong enough (low pKa) can displace carbonic acid from sodium bicarbonate.
Test Procedure (Sodium Bicarbonate Test)
Take a small amount of the organic compound in a clean and dry test tube.
Add fresh aqueous sodium bicarbonate solution to the test tube slowly and carefully.
Observe the mixture for the formation of bubbles or effervescence.
Brisk effervescence due to CO₂ evolution indicates a positive test for carboxylic acids.
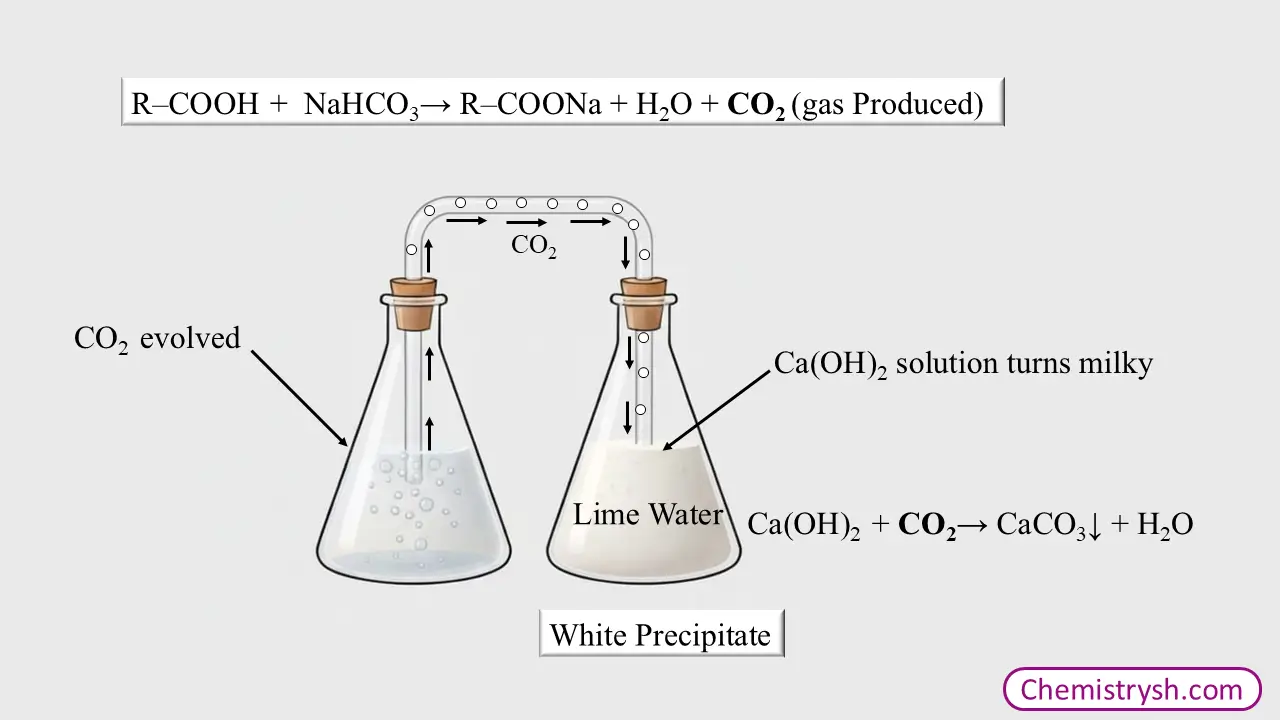
Test for Carbon Dioxide: Limewater Test
During the sodium bicarbonate test, the gas evolved during effervescence is confirmed to be carbon dioxide (CO₂) by performing the limewater test.
Procedure:
- Collect the gas evolved during effervescence.
- Pass it through freshly prepared limewater
.
Observation:
- Limewater turns milky.
Chemical reaction:
Ca(OH)2 + CO2 → CaCO3↓ + H2O
Calcium carbonate is insoluble in water. When carbon dioxide is passed through limewater during the lime water test, insoluble calcium carbonate is formed, which causes the solution to turn milky due to the formation of a white precipitate.
Ca(OH)2 + CO2 → CaCO3↓ + H2O
When excess carbon dioxide is passed through the milky solution, the calcium carbonate reacts further to form calcium bicarbonate, which is soluble in water. As a result, the precipitate dissolves and the milkiness disappears.
CaCO3 + CO2 + H2O → Ca(HCO3)2 (soluble)
Precaution
Carbon dioxide should be passed slowly and only for a short time through limewater. Excess CO₂ must be avoided, otherwise the formed calcium carbonate will convert into soluble calcium bicarbonate, causing the milkiness to disappear and leading to an incorrect observation.
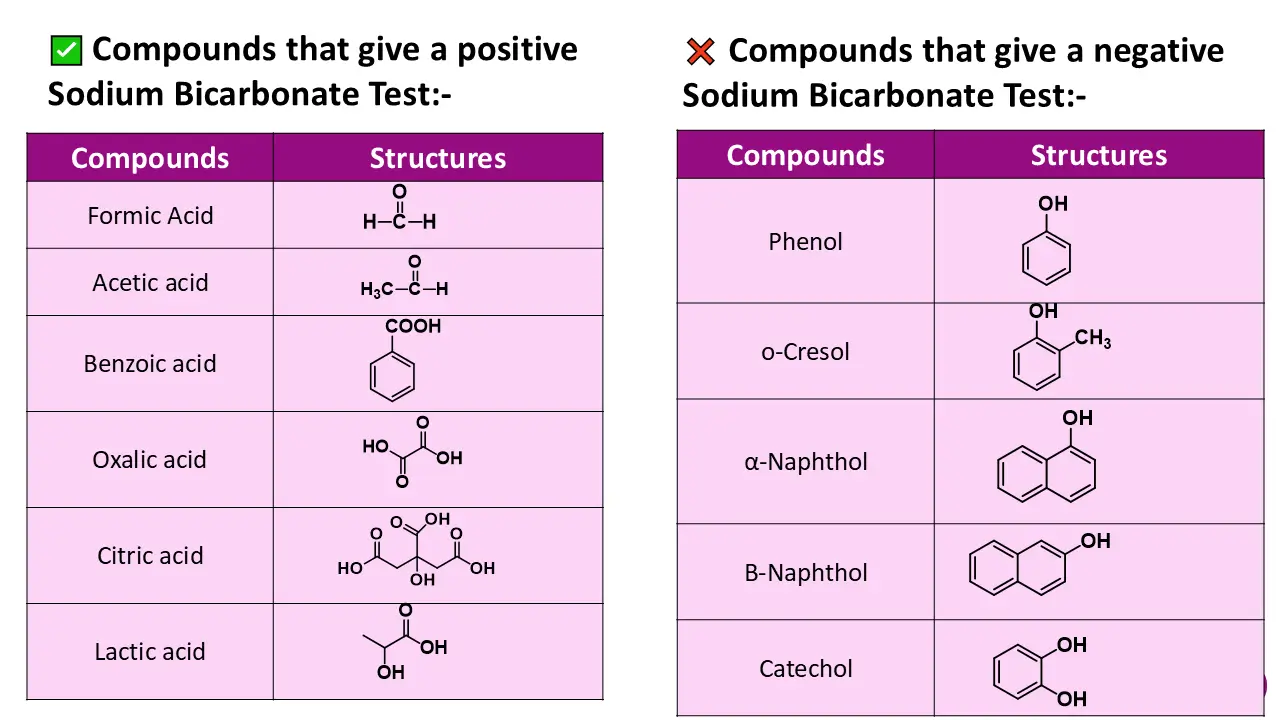
Differentiation Between Carboxylic Acids and Phenols
Carboxylic acids give a positive sodium bicarbonate test due to their stronger acidic nature.
Phenols do not react with sodium bicarbonate because they are weaker acids and cannot liberate carbon dioxide gas. This difference helps in clear differentiation between functional groups. (The pKa of the carboxylic acid ranges from 5 to 6, and that of the phenol is 9 to 10).
Reactions with different Compounds
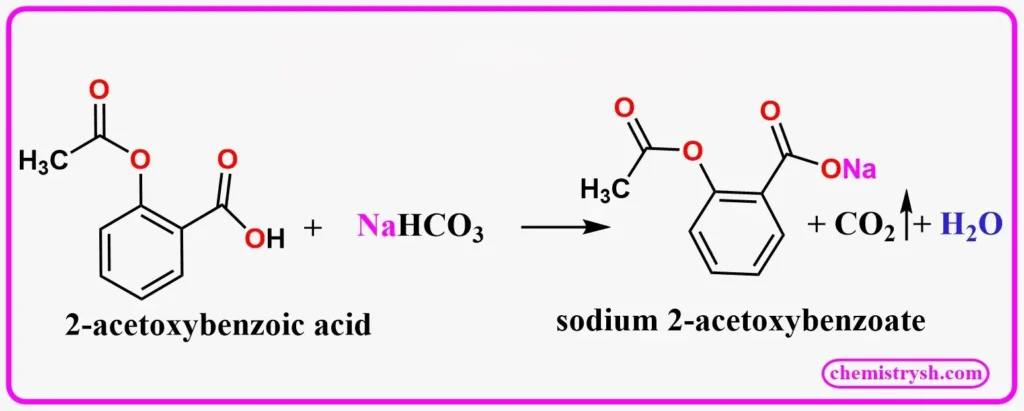
Benzoic acid reacts with sodium bicarbonate (NaHCO₃) to produce sodium benzoate, carbon dioxide gas, and water. The effervescence (bubbling) of CO₂ confirms a positive test.
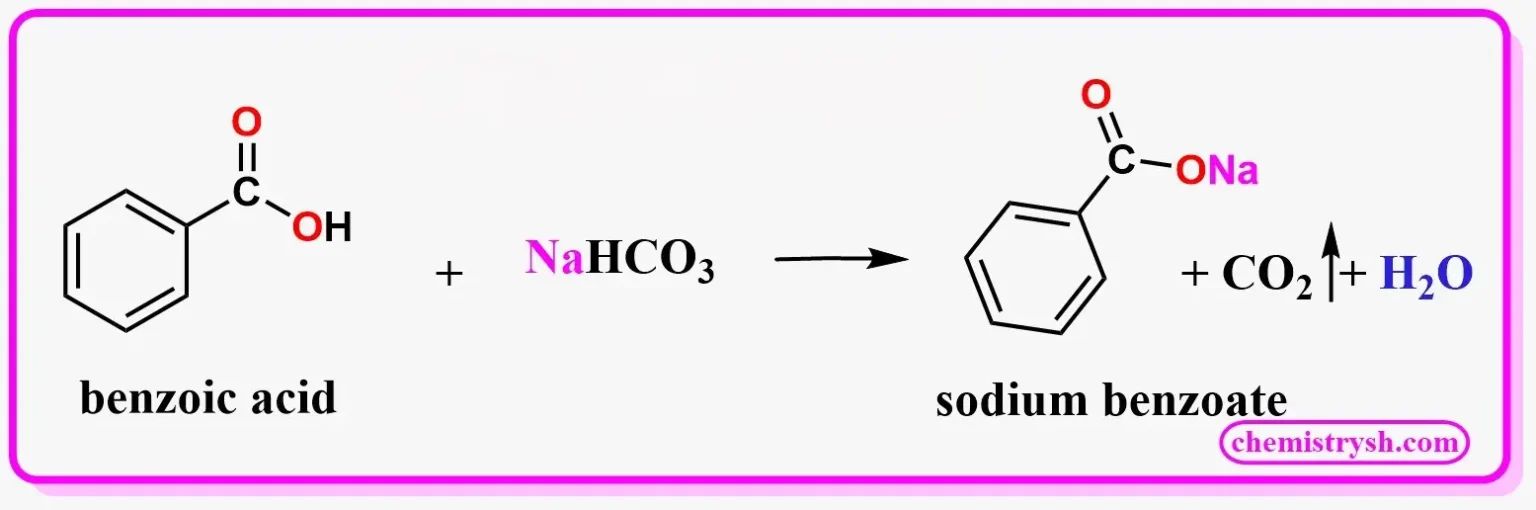
Cinnamic acid, an α,β-unsaturated carboxylic acid, also gives a positive sodium bicarbonate test, forming the sodium salt of cinnamic acid along with CO₂ and H₂O.
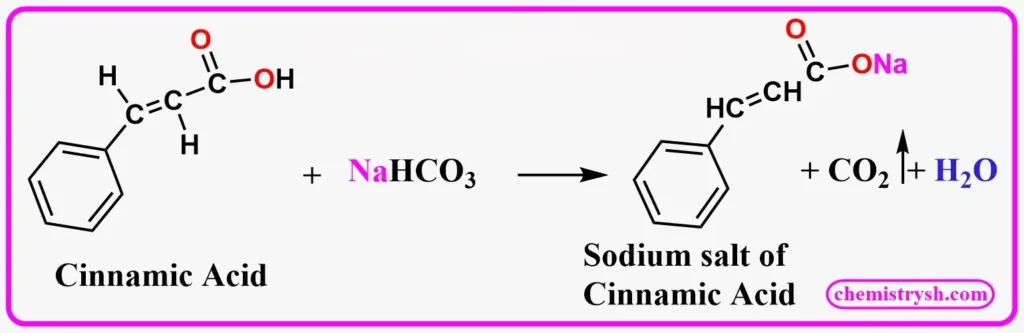
2-Hydroxybenzoic acid (salicylic acid) reacts with NaHCO₃ to give its sodium salt with visible CO₂ evolution, confirming the presence of a carboxylic acid group.
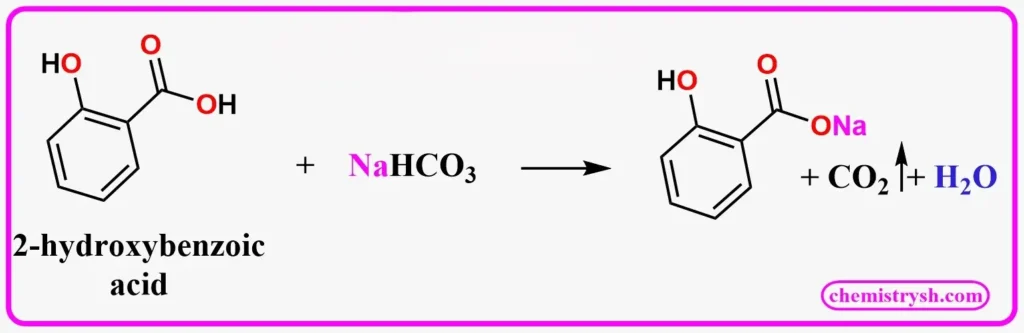
Aspirin (2-acetoxybenzoic acid) reacts with sodium bicarbonate to form sodium 2-acetoxybenzoate. Note that only the –COOH group reacts; the ester group remains intact.
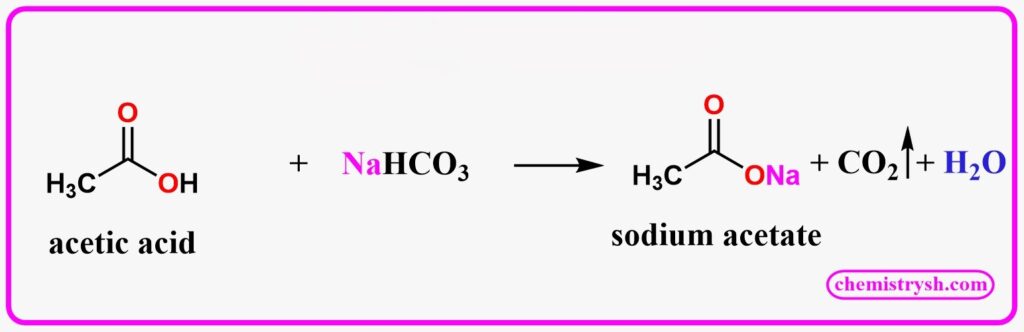
Acetic acid, a simple aliphatic carboxylic acid, reacts with NaHCO₃ to produce sodium acetate, CO₂, and water a classic positive result of the sodium bicarbonate test.
Applications of Sodium Bicarbonate Test
- Used in organic chemistry laboratories for the identification of carboxylic acid functional groups.
- Helps in distinguishing carboxylic acids from phenols and neutral compounds based on acidic strength.
- Commonly employed in educational institutions for teaching organic qualitative analysis.
- Demonstrates the relationship between acidity (pKa) and chemical reactivity.
- Useful in preliminary analysis of unknown organic compounds during laboratory experiments.
- Included in practical examinations and laboratory manuals as a standard confirmatory test.
Viva Voce (Oral Examination) Questions
- What is the principle of the sodium bicarbonate test?
- Why is CO₂ evolved in this test?
- Why do phenols give a negative test?
Multiple Choice Questions
MCQ 1
Sodium bicarbonate test identifies:
A. Alcohols
B. Aldehydes
C. Carboxylic acids
D. Ketones
MCQ 2
2. Gas evolved in the test is:
A. Oxygen
B. Hydrogen
C. Carbon dioxide
D. Nitrogen
MCQ 3
3. Which compound gives effervescence?
A. Phenol
B. Ethanol
C. Acetic acid
D. Aniline
FAQ’s
References
- Vogel’s Textbook of Practical Organic Chemistry
- Shriner et al., The Systematic Identification of Organic Compounds
- Mann and Saunders, Practical Organic Chemistry
- Pavia et al., Introduction to Organic Laboratory Techniques