The solubility of organic compounds is a fundamental concept in organic chemistry that helps in the preliminary identification and classification of unknown substances before confirmatory tests.
What Is Solubility?
Solubility is the ability of a substance (called the solute) to dissolve in another substance (called the solvent) to form a uniform solution at a given temperature.
Solubility tells us how much of a substance can dissolve in a liquid under specific conditions.
The First and Most Important Step in Organic Compound Identification
(How a Chemist Thinks Before Any Test)
Introduction
The identification of an organic compound always begins with one critical decision: determining whether the compound is acidic, basic, or neutral. This decision controls every test that follows and must never be skipped.
This classification is achieved through systematic solubility testing, not guessing and not jumping to confirmatory reactions. A correct solubility decision saves time, chemicals, and prevents wrong conclusions.
How I Teach This Step to Students (Professor’s Guidance)
Before touching any reagent, I tell students one rule:
“Do not ask what the compound is. First ask how it behaves.”
Organic compounds reveal their nature through solubility. If you observe carefully, the compound will guide you toward its correct class of compounds.
Step-by-Step Student Guidance (Must Be Followed)
Step 1: Test Solubility in Water
Take a small amount of the compound and shake it with water.
👉 Do not conclude yet. Only observe.
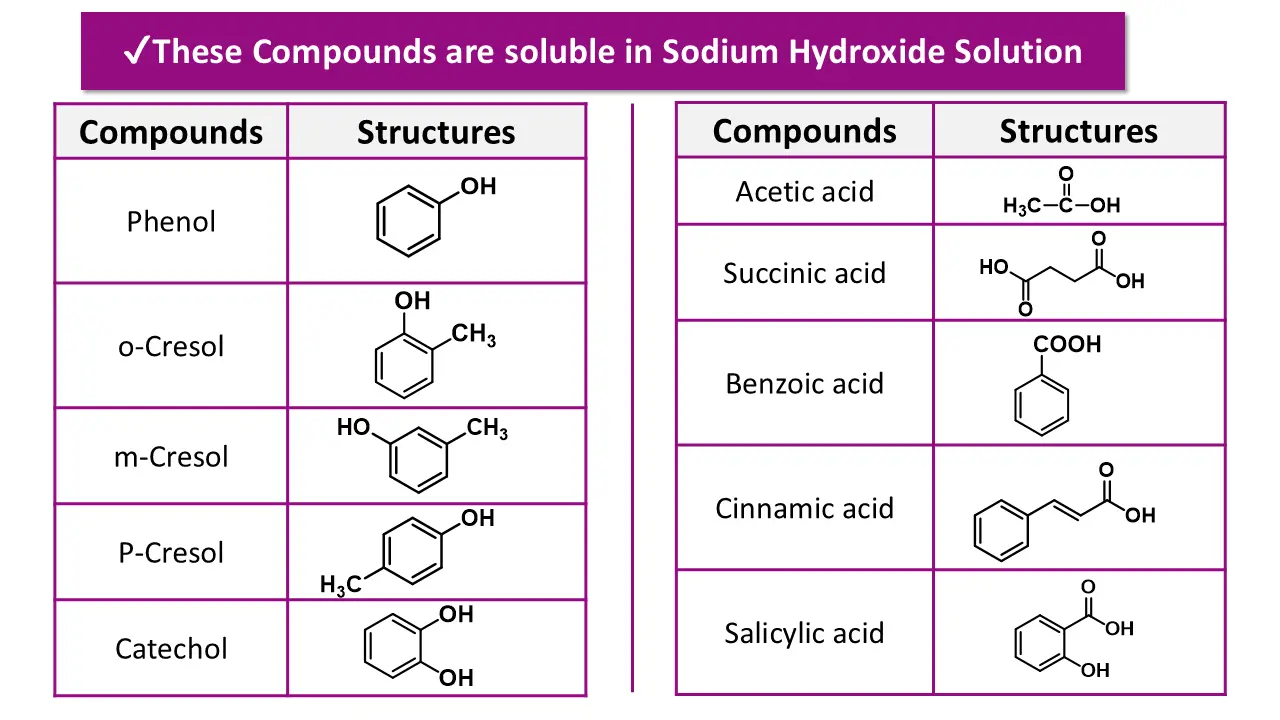
Step 2: Test Solubility in Dilute Sodium Hydroxide
Take a small amount of the compound and shake it Add dilute NaOH to a fresh portion of the compound.
👉 Record solubility. Do not name the functional group yet.
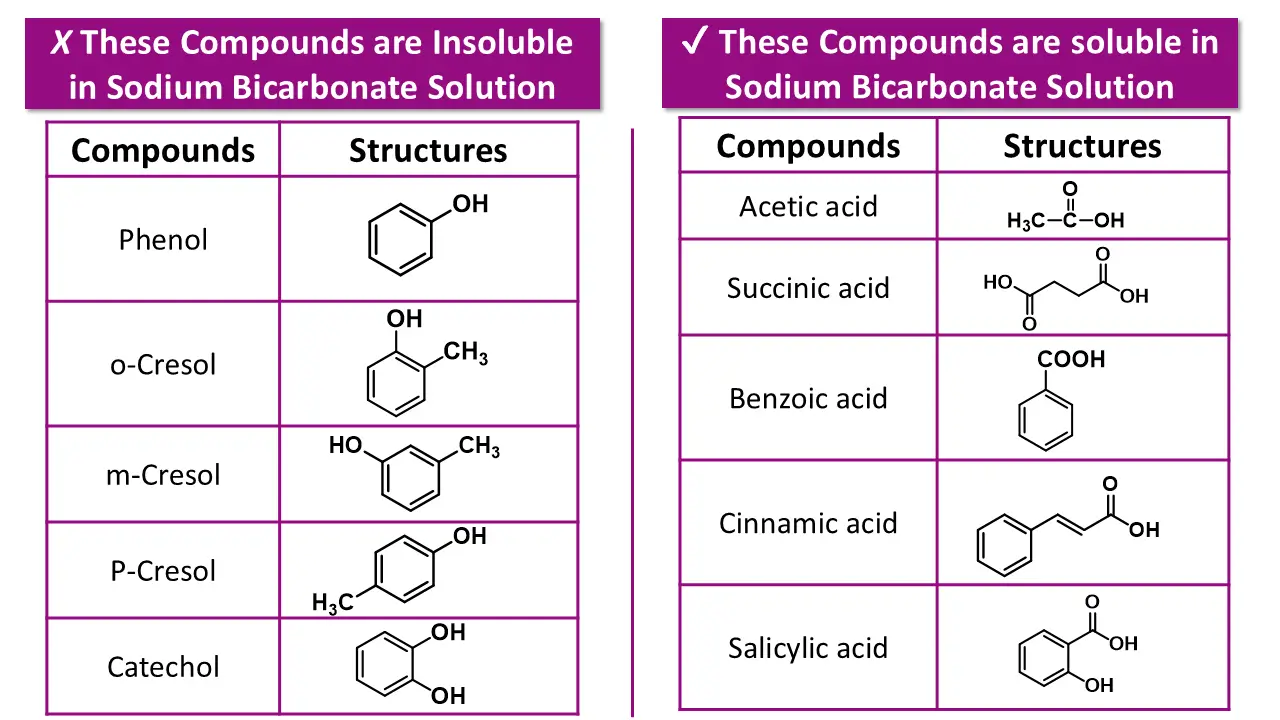
Step 3: Test Solubility in Sodium Bicarbonate
Take a small amount of the compound and shake iNow test with sodium bicarbonate solution.
👉 This step confirms acidity strength, not identity.
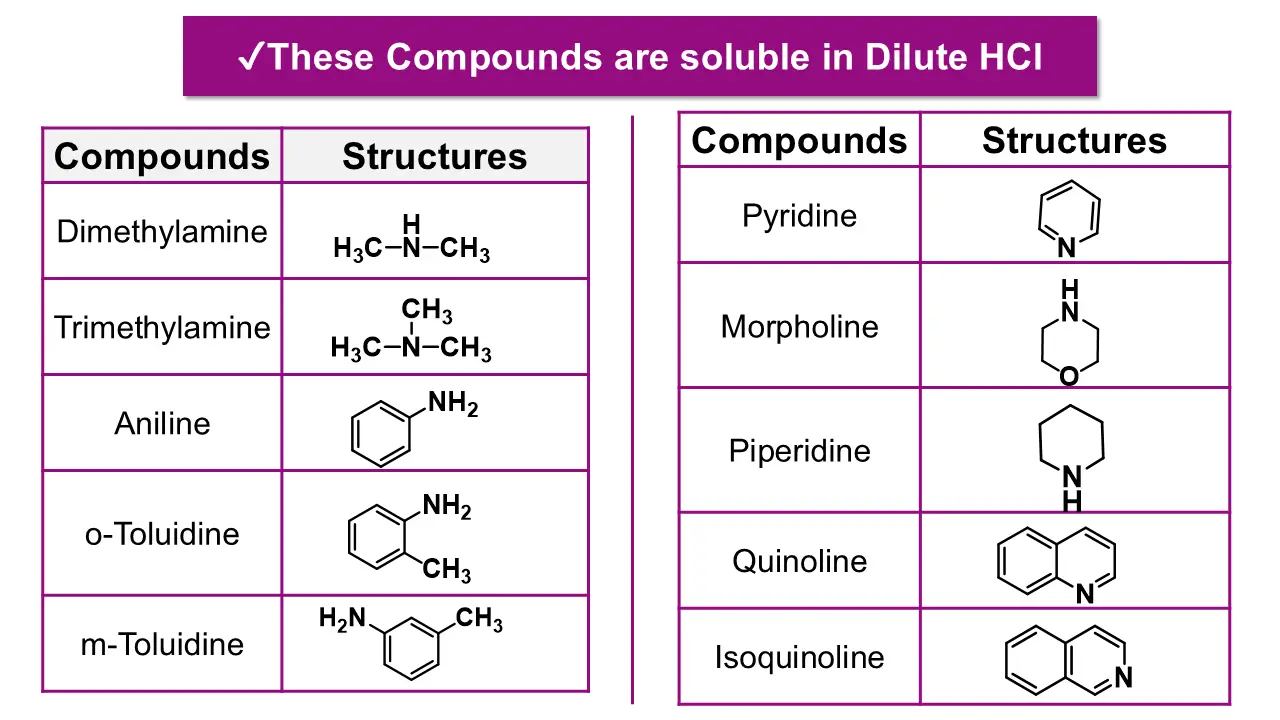
Step 4: Test Solubility in Dilute Hydrochloric Acid
Test the compound with dilute HCl.
👉 This separates bases from neutral compounds.
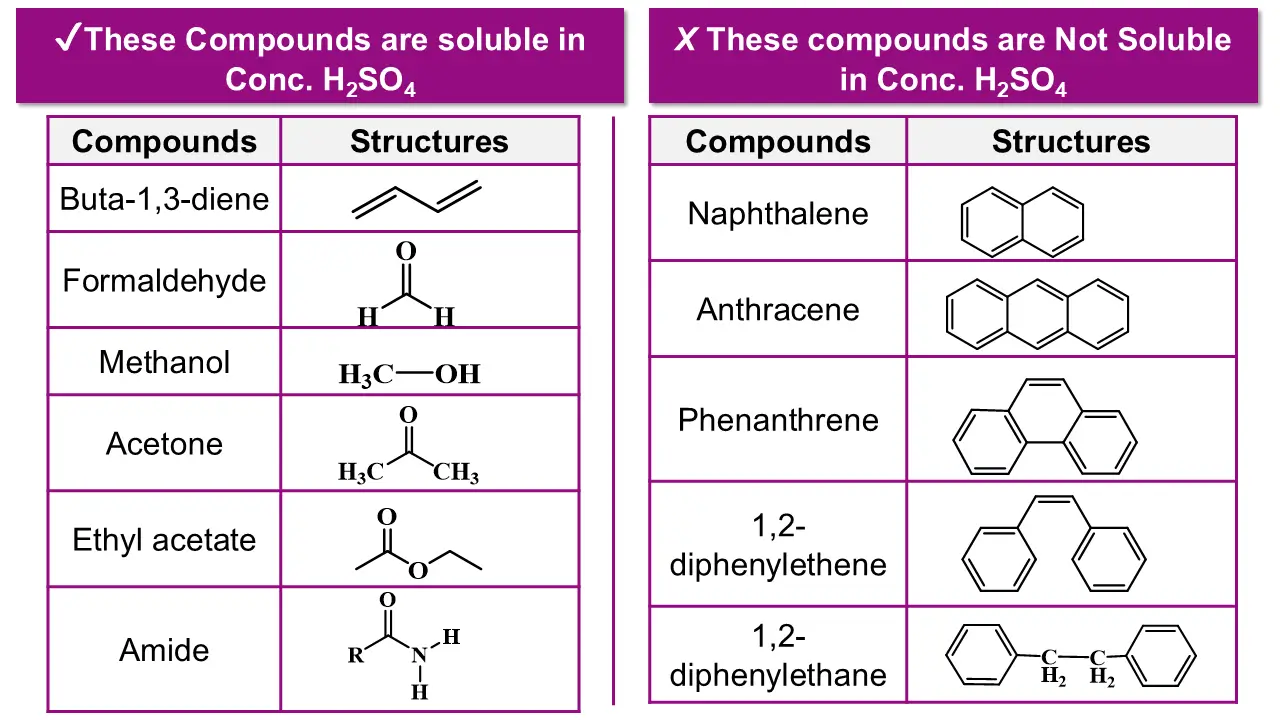
Step 5: Test Solubility in Concentrated Sulfuric Acid
Finally, test with concentrated H₂SO₄ (with caution).
👉 This test is diagnostic, not confirmatory.
What I Repeatedly Tell Students
- Do not jump to conclusions
- Do not skip solubility tests
- Do not perform confirmatory tests blindly
- One wrong decision here ruins the entire analysis
A chemist thinks stepwise, not emotionally.
Did you know?
Many experienced chemists can correctly predict the functional group of an unknown compound only from its solubility behaviour, without performing a single confirmatory reaction.
Educational Outcome for Students
After completing solubility testing, the student should be able to:
- Decide whether the compound is acidic, basic, or neutral
- Eliminate impossible functional groups
- Choose the correct confirmatory tests
- Avoid unnecessary and misleading reactions
Stepwise Scheme for Identification Using Solubility
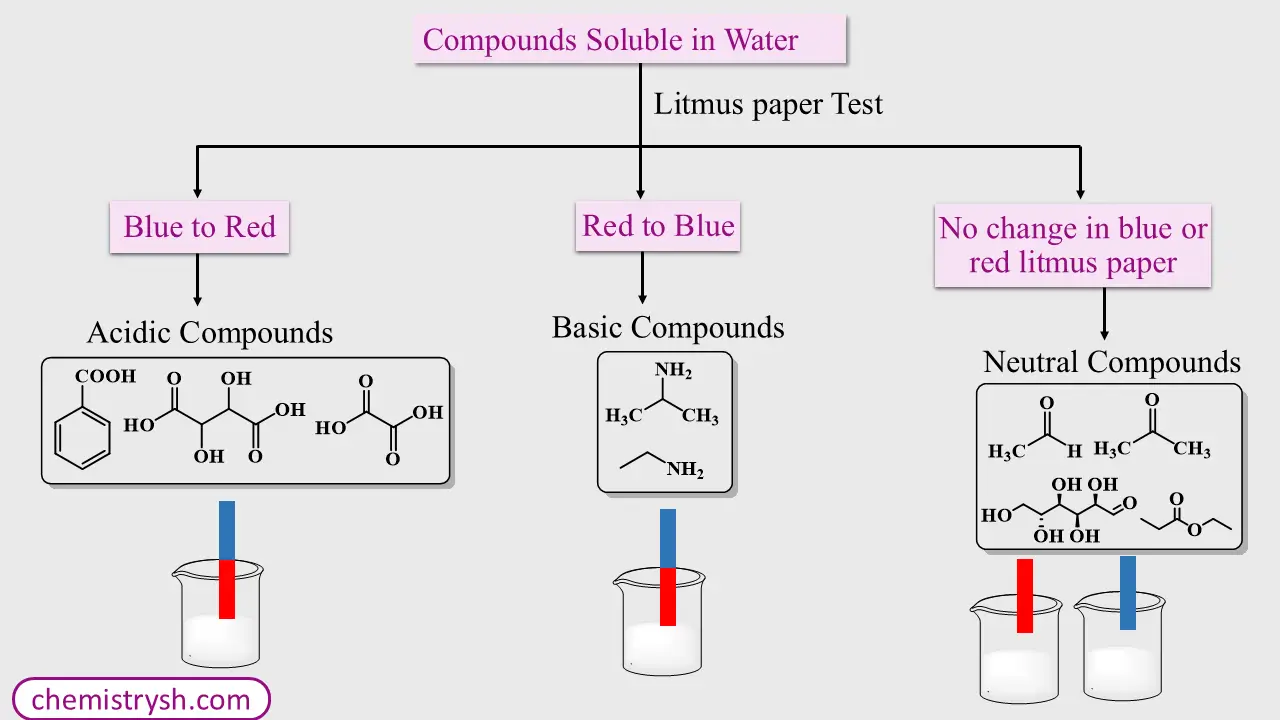
Step 1: Solubility in Water
Step 1: Solubility in Water
- A small amount of the organic compound is shaken with distilled water.
(a) Compound Soluble in Water
- Perform litmus paper test:
- Blue litmus turns red → Acidic compound
- Red litmus turns blue → Basic compound
- No change → Neutral compound
Water-soluble compounds are usually low molecular weight acids, bases, or neutral polar compounds.
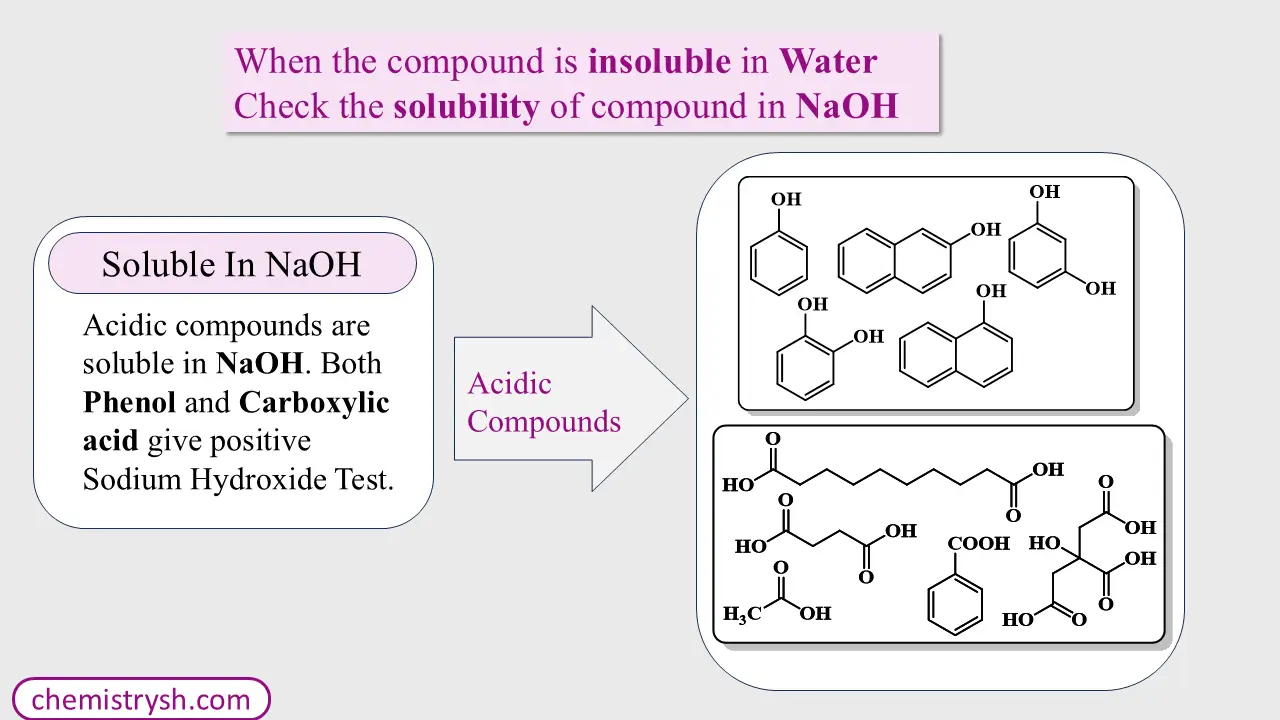
Step 2: Solubility in Sodium Hydroxide (NaOH)
If the compound is insoluble in water, treat it with aqueous NaOH.
(a) Soluble in NaOH
- Indicates the compound is acidic in nature.
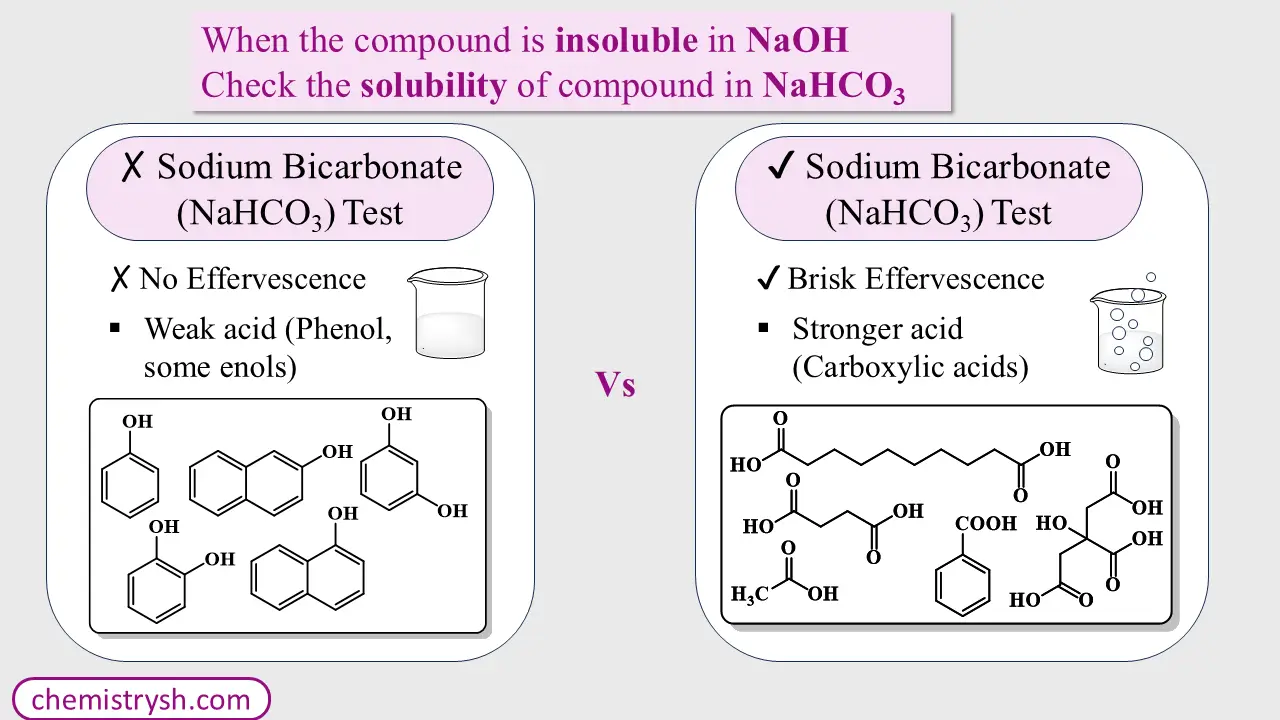
Solubility in Sodium Bicarbonate (NaHCO₃)
(b) Now perform sodium bicarbonate (NaHCO₃) test:
Step 3: Solubility in Hydrochloric Acid (HCl)
If the compound is insoluble in both water and NaOH, treat it with dilute HCl.
(a) Soluble in HCl
Further confirmed by specific tests for primary, secondary, or tertiary amines
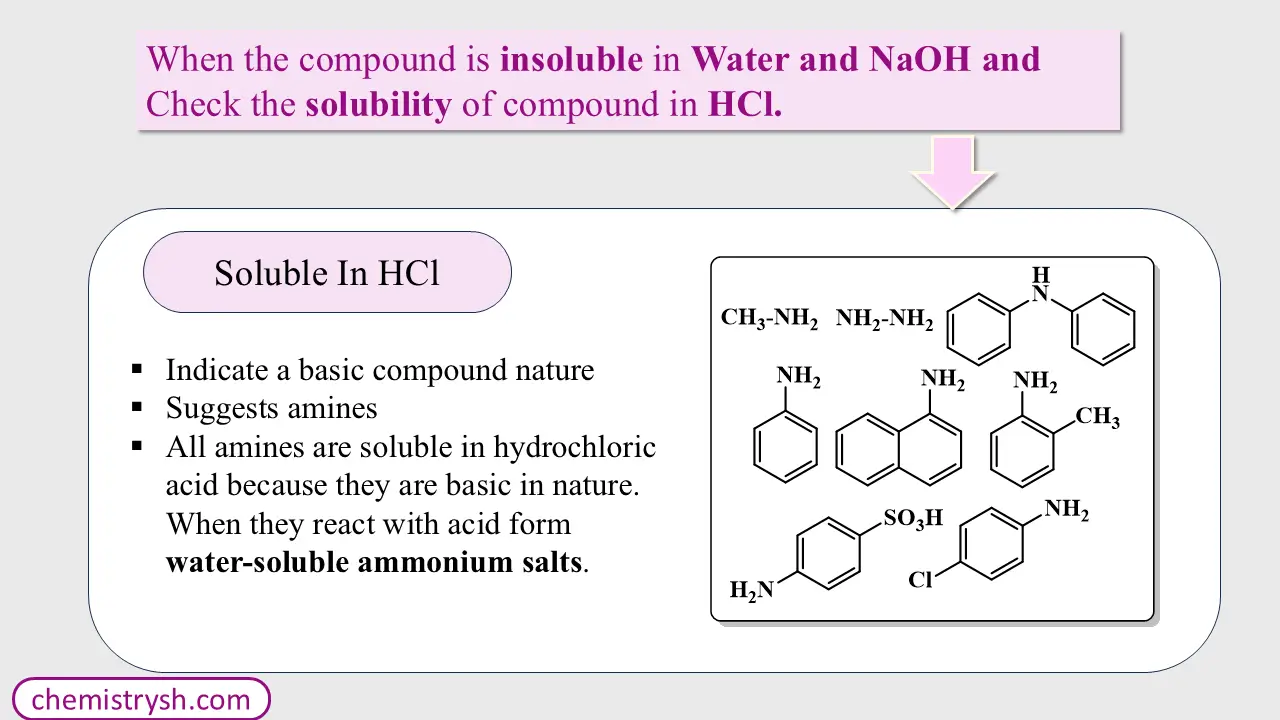
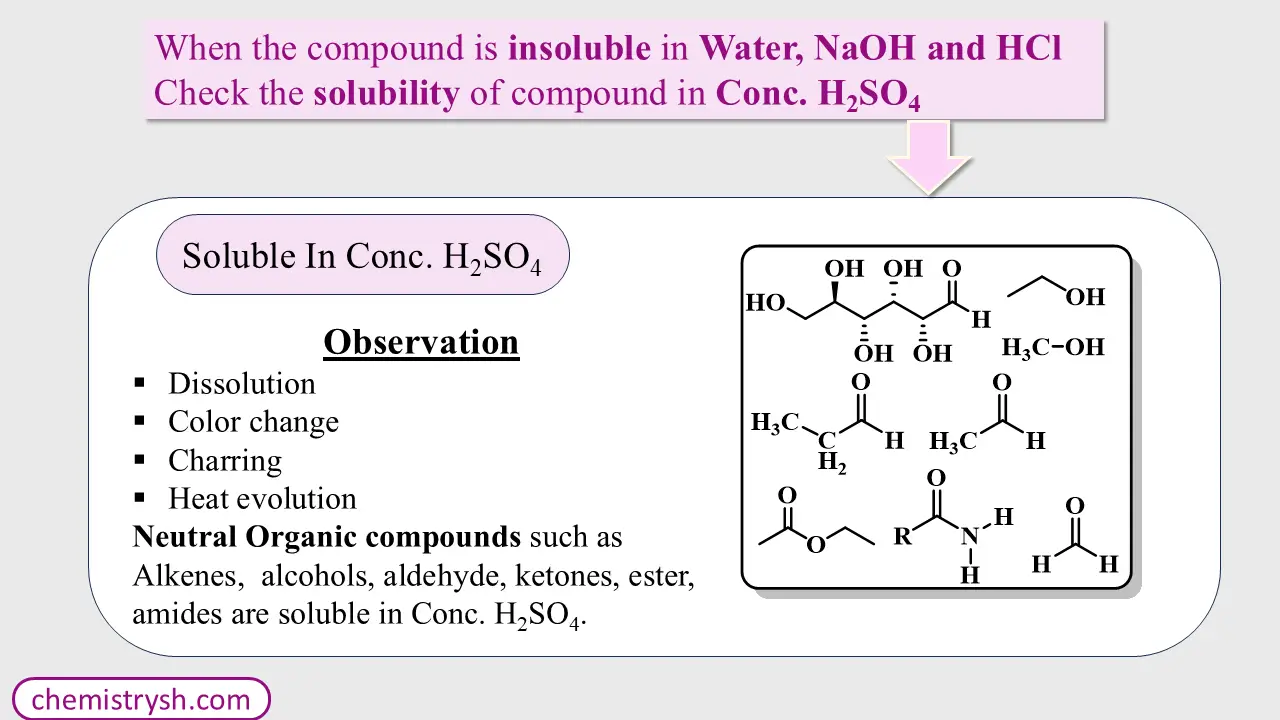
Step 4: Reaction with Concentrated Sulfuric Acid (H₂SO₄)
If the compound is insoluble in water, NaOH, and HCl, treat it with cold concentrated sulfuric acid.
Possible observations:
- Dissolution
- Color change
- Heat evolution
- Charring
These observations indicate reactive neutral compounds, such as: Alkyl halides, Alcohols, Aldehydes, Ketones and Alkenes.
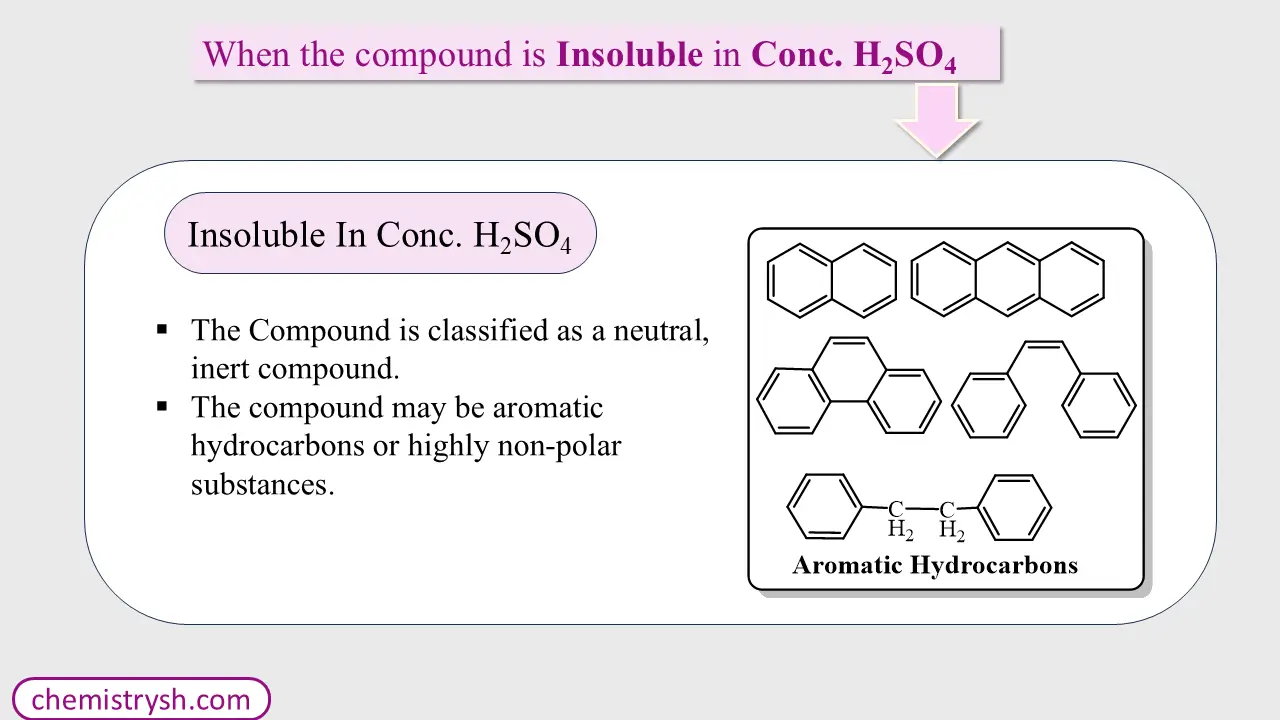
Step 5: No Reaction with Concentrated Sulfuric Acid
If there is no reaction even with concentrated H₂SO₄:
Usually aromatic hydrocarbons or saturated hydrocarbons
The compound is classified as a neutral, inert compound.
Summary Table: Solubility of Organic Compounds
|
Solubility Behavior |
Inference |
|
Water soluble + acidic |
Low molecular weight acids |
|
Soluble in NaOH only |
Weak acid (phenol) |
|
Soluble in NaOH + effervescence with NaHCO₃ |
Carboxylic acid |
|
Soluble in HCl |
Basic compound (amine) |
|
Reacts with conc. H₂SO₄ |
Reactive neutral compound |
|
No reaction with conc. H₂SO₄ |
Aromatic hydrocarbon |

Applications of Solubility Method
- Rapid preliminary classification of unknown compounds
- Reduces number of confirmatory tests
- Widely used in undergraduate chemistry laboratories
- Helps in systematic qualitative organic analysis
Viva questions
- What is the aim of solubility tests in organic chemistry?
- Why is litmus paper used for water-soluble compounds?
- What is the role of NaOH in solubility tests?
- How can carboxylic acids be distinguished from phenols?
- Why are aromatic hydrocarbons insoluble in water?
- What happens when an amine reacts with HCl?
- Why is H₂SO₄ used for neutral compounds?
Multiple Choice Questions
MCQ 1
1. Aromatic hydrocarbons are:
A. Polar
B. Water soluble
C. Highly reactive
D. Insoluble in acids and bases
MCQ 2
2. Which compound shows effervescence with NaHCO₃?
A. Alcohol
B. Phenol
C. Carboxylic acid
D. Amine
MCQ 3
3. A compound soluble in HCl but insoluble in NaOH is:
A. Acidic
B. Neutral
C. Basic
D. Aromatic
MCQ 4
4. Which test is used to identify acidic compounds?
A. HCl test
B. NaOH test
C. H₂SO₄ test
D. Litmus only
MCQ 5
5. Neutral compounds are identified by solubility in:
A. NaOH
B. NaHCO₃
C. HCl
D. H₂SO₄
MCQ 6
6. Which form of ferric chloride is used in this test?
A. Solid FeCl₃
B. Alcoholic FeCl₃
C. Aqueous neutral FeCl₃
D. Concentrated FeCl₃
FAQ’s
References
- Science Direct Journal
- Furniss, B. S., Hannaford, A. J., Smith, P. W. G., & Tatchell, A. R. (1989). Vogel’s Textbook of Practical Organic Chemistry (5th ed., pp. 106–125). Longman Scientific & Technical.
- Shriner, R. L., Fuson, R. C., Curtin, D. Y., & Morrill, T. C. (2004). The Systematic Identification of Organic Compounds (8th ed., pp. 1–32). John Wiley & Sons.
- Mann, F. G., & Saunders, B. C. (1970). Practical Organic Chemistry (4th ed., pp. 215–230). Longman.
- Ahluwalia, V. K., & Aggarwal, R. (2008). Comprehensive Practical Organic Chemistry (pp. 15–28). Universities Press.